

By Jasmin Cordy
March 15, 2017

Butylated hydroxytoluene (BHT) is commonly used in cosmetic formulations as an antioxidant. In addition to personal care products and makeup, it is also widely used in plastics and foods. BHT (or 2,6-di-tert-butyl-p-cresol) is a white to yellowish crystalline solid that prevents the oxidation of fats and oils, and helps to extend a product's shelf-life. It is produced by the reaction of p-cresol with isobutylene and sulfuric acid.1 The key function of BHT is that of a stabilizer. It inhibits the degradation of fats and oils through its antioxidant action and prevents rancidity. BHT is also a food additive, which is approved by the Food and Drug Administration, and is generally recognized as safe (GRAS).2 In cosmetic formulations, BHT is often used at concentrations ranging from 0.0001% to 0.5%.3
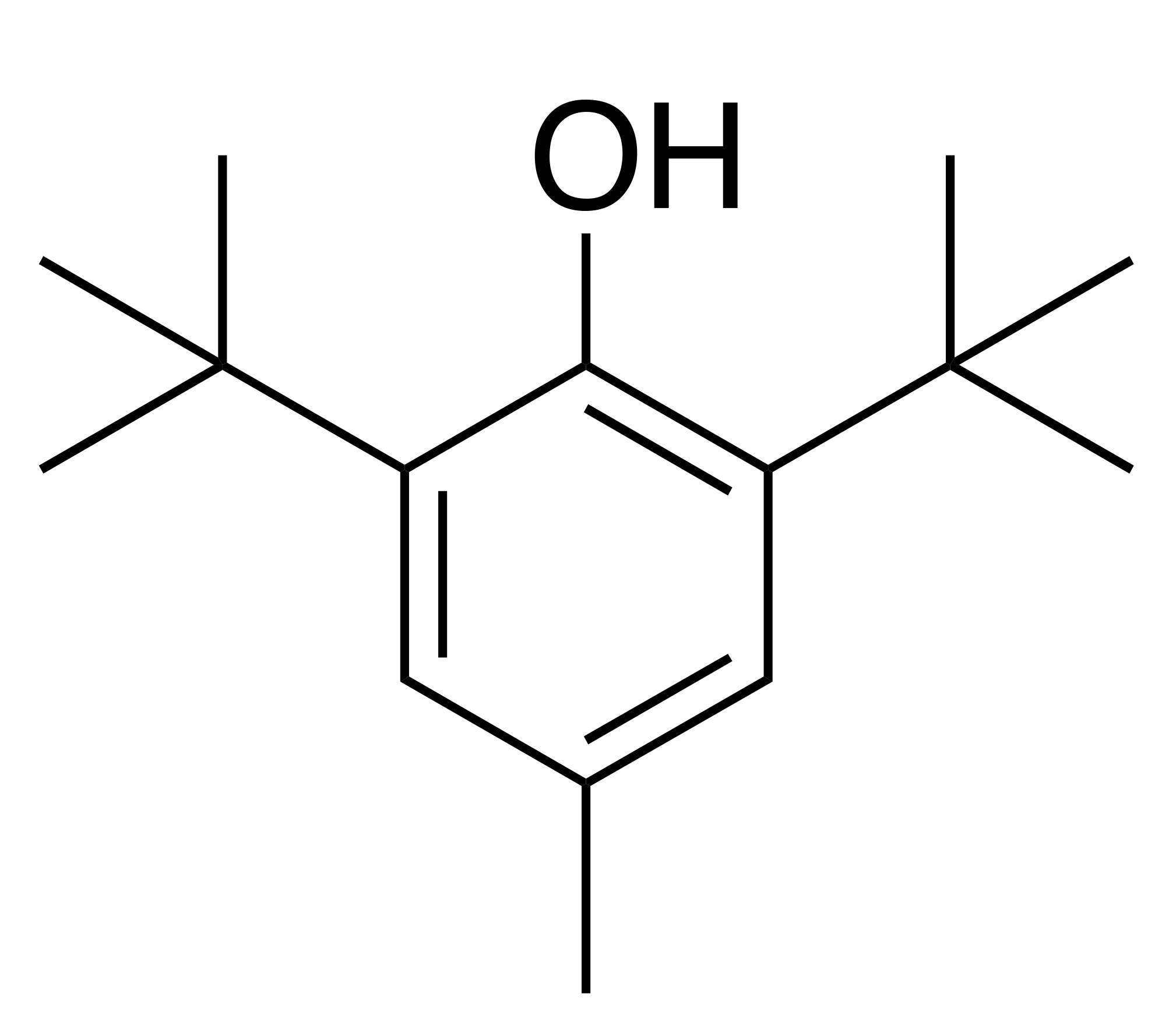
Figure 1: Molecular structure of BHT.
Consumers seek safe, natural ingredients for skin, hair, body, and food products. Nowadays, more consumers read and research the information found on packaging labels including ingredient listings. There has been increased concern and caution amongst consumers with the use of synthetic additives in foods and cosmetics. BHT has been questioned for safety and efficacy due to consumers' skepticism of additives of synthetic origin. In fact, a major manufacturer of cereals volunteered to remove BHT from its cereals due to negative concerns of consumers.4 Based on toxicological studies it was concluded that BHT may produce toxicity in high oral doses and can cause adverse effects in some organs using animal models.5,6
Although there are some concerns about BHT, scientific research maintains that when used at low concentrations BHT is safe. Long-term use studies have substantiated the overall safety and efficacy of BHT.7 In skin permeation studies, it was demonstrated that BHT has the ability to penetrate the skin; however, the amount absorbed does not appear to pass through the top layer of the skin, and it has been determined to be safe for use in cosmetics.3 It was also concluded that application of BHT to the skin does not produce appreciable systemic levels of the molecule, and it does not result in widespread irritation, sensitization, or photosensitization. In addition to its antioxidant protection of cosmetic preparations, BHT provides positive benefits for the skin. In one study, it was shown that BHT may have photoprotective properties and possibly impede photocarcinogenesis when used in conjunction with carotenoids, such as beta-carotene.8
Although BHT is still widely used, some companies are searching for alternatives with good toxicological and ecological profiles. Some possible alternatives are BHT analogues that offer strong stabilizer benefits, such as tocopherols and synthetic analogues.9 Another option is to promote BHT as a naturally sourced antioxidant. It has been shown that BHT can be produced in freshwater aquatic plants, such as phytoplankton.10
BHT has been in use for a long time in consumer products. Its safety profile has led to its inclusion in many products resulting in extensive use. Scientific data supports the safety of BHT for the consumer. BHT is a mainstay of the cosmetics and food industry and is beneficial for the safe preservation of cosmetics, such as cleansers, lotions, make-up, and hairstyling products. Further research could discover even more beneficial uses of BHT.
References
1. H. Fiege, H.-W. Voges, T. Hamamoto, S. Umemura, T. Iwata, H. Miki, Y. Fujita, H.-J. Buysch, D. Garbe, and W. Paulus, “Phenol Derivatives” in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH: Weinheim, Germany (2002); doi:10.1002/14356007.a19_313.
2. U.S. Food & Drug Administration, Select Committee on GRAS Substances (SCOGS) Opinion: Butylated Hydroxytoluene (BHT).
3. R.S. Lanigan and T.A. Yamarik, Final report on the safety assessment of BHT, Int. J. Toxicol., 21 Suppl 2, 19-94 (2002).
4. General Mills, BHT statement, February 5, 2015.
5. H. Verhagen, H.H. Beckers, P.A. Comuth, L.M. Maas, F. ten Hoor, P.T. Henderson, and J.C. Kleinjans,
Disposition of single oral doses of butylated hydroxytoluene in man and rat, Food Chem. Toxicol., 27, 765-772 (1989).
6. C.J. Powell, J.C. Connelly, S.M. Jones, P. Grasso, and J.W. Bridges, Hepatic responses to the administration of high doses of BHT to the rat: their relevance to hepatocarcinogenicity, Food Chem. Toxicol., 24, 1131-1143 (1986).
7. G.M. Williams, M.J. Iatropoulos, and J. Whysner, Safety assessment of butylated hydroxyanisole and butylated hydroxytoluene as antioxidant food additives, Food Chem. Toxicol., 37, 1027-1038 (1999).
8. H.S. Black and M.M. Mathews-Roth, Protective role of butylated hydroxytoluene and certain carotenoids in photocarcinogenesis, Photochem. Photobiol., 53 , 707-716 ( 1991).
9. W.A. Yehye, N. Abdul Rahman, A.A. Alhadi, H. Khaledi, N.S. Weng, and A. Ariffin, Butylated hydroxytoluene analogs: synthesis and evaluation of their multipotent antioxidant activities, Molecules, 17, 7645-7665 (2012).
10. B. Babu and J.T. Wu, Production of natural butylated hydroxytoluene as an antioxidant by freshwater phytoplankton, J. Phycol., 44, 1447-1454 (2008).