

by Roger L. McMullen and Gopinathan K. Menon
The epidermis is a stratified, constantly renewing epithelium that undergoes a process known as differentiation, which is also known as keratinization or cornification. It is comprised of several morphological layers that are classified according to the functional and/or structural state of the keratinocyte, the predominant cell type in these layers. The various layers of the epidermis consist of the stratum basale, stratum spinosum, stratum granulosum, and stratum corneum. In addition to keratinocytes, the epidermis also houses the melanocytes and sentinel cells (Langerhans cells).
The process of keratinization consists of the loss of the ability of keratinocytes to proliferate ;along with several key transformations in the cellular and morphological structure. Keratinocytes change form from columnar-shaped cells in the stratum basale layer to flat polyhedral shaped cells filled with keratin in the stratum corneum. Cellular organelles, such as the mitochondrion, Golgi apparatus, nucleus, etc., are lost and new organelles are formed as part of the preparation for the cornified layer (stratum corneum).
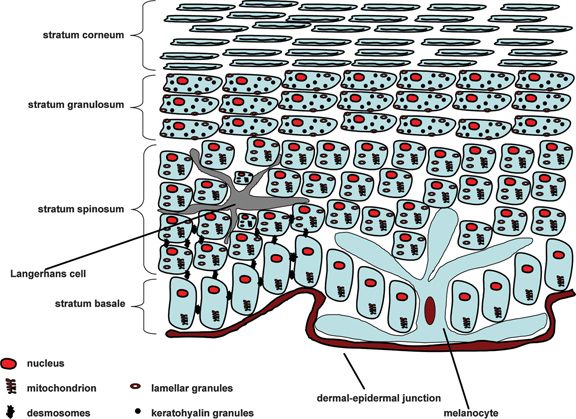
Figure 2. Diagram of the epidermis. Copyright (2013) Allured Publishing, Reprinted with permission from R. McMullen, Antioxidants and the Skin, Allured Books: Carol Stream, IL (2013).
In addition, in the lower parts of the epidermis, lipids found in normal viable cells can be found, such as phospholipids, while these diminish in the upper layers of the epidermis, which is mostly composed of ceramides. Another hallmark of keratinization is the structural transformation of the plasma membrane from a fluid phospholipid bilayer to a tough, protein rich cornified cell envelope. Finally, dehydration occurs with a mostly aqueous environment present in the lower layers of the epidermis and a predominantly dry state in the stratum corneum.
A number of different keratins are expressed in each of the layers, which allow for the transformation of the keratinocyte as it traverses through the various layers of the epidermis. The outermost layer is the stratum corneum, which serves as the barrier between the viable layers of the skin and the outside world.
Keratin protein expression is a hallmark of the keratinocyte activity. Depending on the needs of the cell, in terms of agility, flexibility, rigidness, etc., certain keratins are expressed so that the cell can modify its shape and carry out its function.
Keratins expressed in the skin belong to the acidic or basic class and are paired together. They form intermediate filaments, which are molecular assemblies of long filaments that have the following hierarchical structure: two alpha-keratin chains twist together to form a coiled-coil; two coiled-coils pair together forming a protofilament; two protofilaments associate to form a protofibril; and four protofibrils aggregate to form an intermediate filament (see Figure 3).
The intermediate filaments are approximately 8–10 nm in diameter and have a molecular weight of 40–70 KDa. In most cases, the intermediate filaments extend through the cytoskeleton of the cell and connect the nuclear envelope with desmosomes and hemidesmosomes. Desmosomes are a structural molecular assembly that provides junction points between neighboring keratinocytes, essentially keeping the cellular components of the epidermis bound together. Hemidesmosomes connect basale cell keratinocytes with the underlying dermal-epidermal junction.
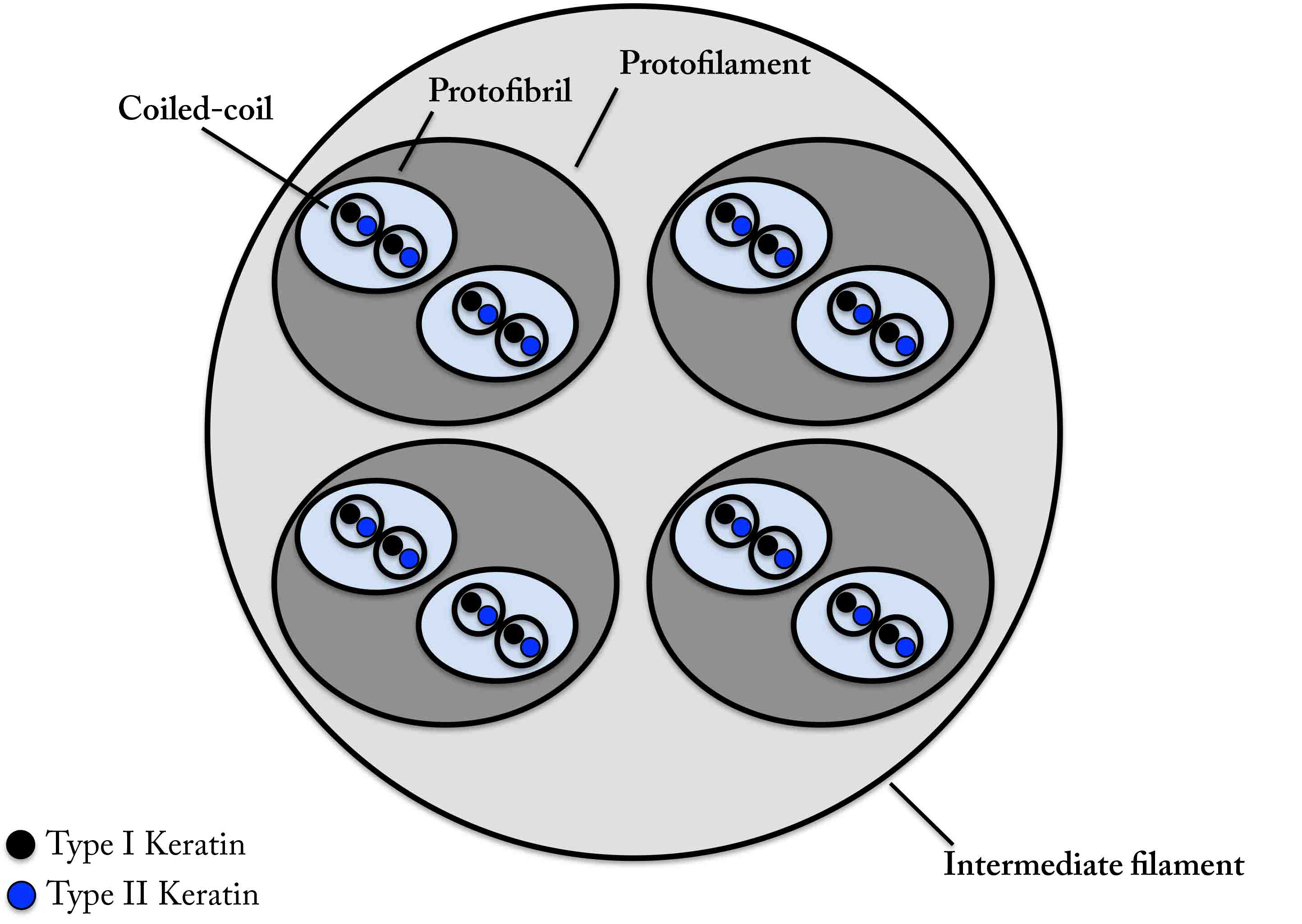
Figure 3. Molecular architecture of the intermediate filament.
As mentioned earlier, in addition to keratinocytes, which make about 90–95% of the cells of the epidermis, there are also melanocytes, Langerhans cells, and Merkel cells. Melanocytes are dendritic cells that produce melanosomes, which are organelles that contain melanin. The long arms of the melanocytes extend throughout the basal layer of the epidermis and deposit melanosomes around the cell nuclei of the keratinocytes (a kind of microparsol) thereby protecting the genetic material of the cell from UV radiation. Langerhans cells are also dendritic cells that essentially serve as sentinels to the immune system. They are antigen-presenting cells, which are able to migrate from the epidermis to nearby lymph tissue and present antigens to other cells of the immune system, eventually leading to the fabrication of antibodies. Finally, Merkel cells are mechanoreceptor cells—mostly located in the stratum basale layer—associated with the nervous system and are responsible for tactile sensation or light touch.
Stratum Basale
As the deepest layer of the epidermis, the stratum basale (also referred to as the stratum germinativum) is located at the interface of the epidermis with the dermal-epidermal junction. It contains columnar-shaped keratinocyte cells. Half of these cells remain in the basale layer and serve as mother cells (mitotically dividing stem cells) while the other half (the daughter cells) migrate upwards through the various strata of the epidermis while undergoing the process of differentiation. In this layer, the keratinocytes most strongly express the K5 and K14 pair, which provides the cells with a more flexible cytoskeleton allowing them to easily undergo mitosis and to migrate.
Stratum Spinosum
Also known as the prickle cell or spinous layer, the stratum spinosum is the layer just above the stratum basale. In this layer, there is an abundance of desmosomes connecting the keratinocytes together. During the histological preparation of ex vivo skin sections the tissue shrinks; however, all of the junction points between the keratinocytes (from the desmosomes) remain, leading to the characteristic spinous appearance of this layer. Lamellar granules (also known as lamellar bodies or Odland bodies) begin to appear in this layer, although their function is not carried out until they arrive at the interface of the stratum granulosum and stratum corneum. These granules are membrane-bound organelles that contain precursors of stratum corneum lipids as well as hydrolases, protease inhibitors, and antiomicrobial peptides.1
Stratum Granulosum
As a major transition region in the epidermis, the stratum granulosum is only 2–3 layers of cells thick. Many important events occur in this layer, including the activity of lamellar bodies and the appearance of keratohyalin granules. Overall, there is a drastic transformation in this layer from a viable to cornified cell, which undergoes a form of apoptosis, essentially destroying many of the keratinocyte’s cellular organelles.
As already mentioned, it is at the interface of the stratum granulosum and stratum corneum where the lipid precursors and enzymes of the lamellar bodies are secreted into intercellular space, eventually forming the lipid matrix of the stratum corneum.
The keratohyalin granules are also essential for skin homeostasis. They contain profilaggrin, keratin intermediate filaments, involucrin and loricrin (components of the cornified envelope). Profilaggrin is a high molecular weight (> 220 KDa) protein structure composed of repeating subunits of filaggrin (37 KDa), a histidine-rich protein.2 One of the primary functions of filaggrin is to aggregate keratin intermediate filaments to form microfibrils. Filaggrin is also very important inasmuch that it is broken down in the stratum corneum into amino acid fragments that constitute the natural moisturizing factor.
As briefly mentioned, keratohyalin granules also contain loricrin, an important structural protein that forms part of the cornified envelope—the tough, resistance membrane surrounding corneocytes. Loricrin is characterized by its glycine, serine, and cysteine residues as well as its involvement in forming isodipeptide crosslinks.3 In addition, involucrin (68 KDa)—another component of the cornified envelope—also appears in the stratum granulosum.4
To summarize, a number of key events take place at the interface between the stratum corneum and stratum corneum. Keratohyalin granules and lamellar bodies bring essential molecules to the interface that are key for the formation of the stratum corneum. Keratin intermediate filaments aggregated together within the cell to form microfibrils. Keratinocyte organelles and cytoplasm are lost leading to the formation of the corneocyte. The cornified cell envelope—a tough predominantly protein membrane—is formed underneath the plasma membrane.
Stratum Lucidum
Histologically, this strata is seen as a clear layer of cells that characterize the epidermis of the palm and soles, but not elsewhere in the body skin.
Stratum Corneum
The stratum corneum is a barrier to water loss, enabling a fairly aqueous organism to live in an arid environment. It also serves as a barrier to permeation of foreign substances. Principally, the stratum corneum is composed of corneocytes (differentiated keratinocytes) embedded in a lipid matrix that primarily consists of ceramides, free fatty acids, and cholesterol, in a specific ratio. In addition, there are lipids (ceramides) that are covalently attached to the corneocytes.
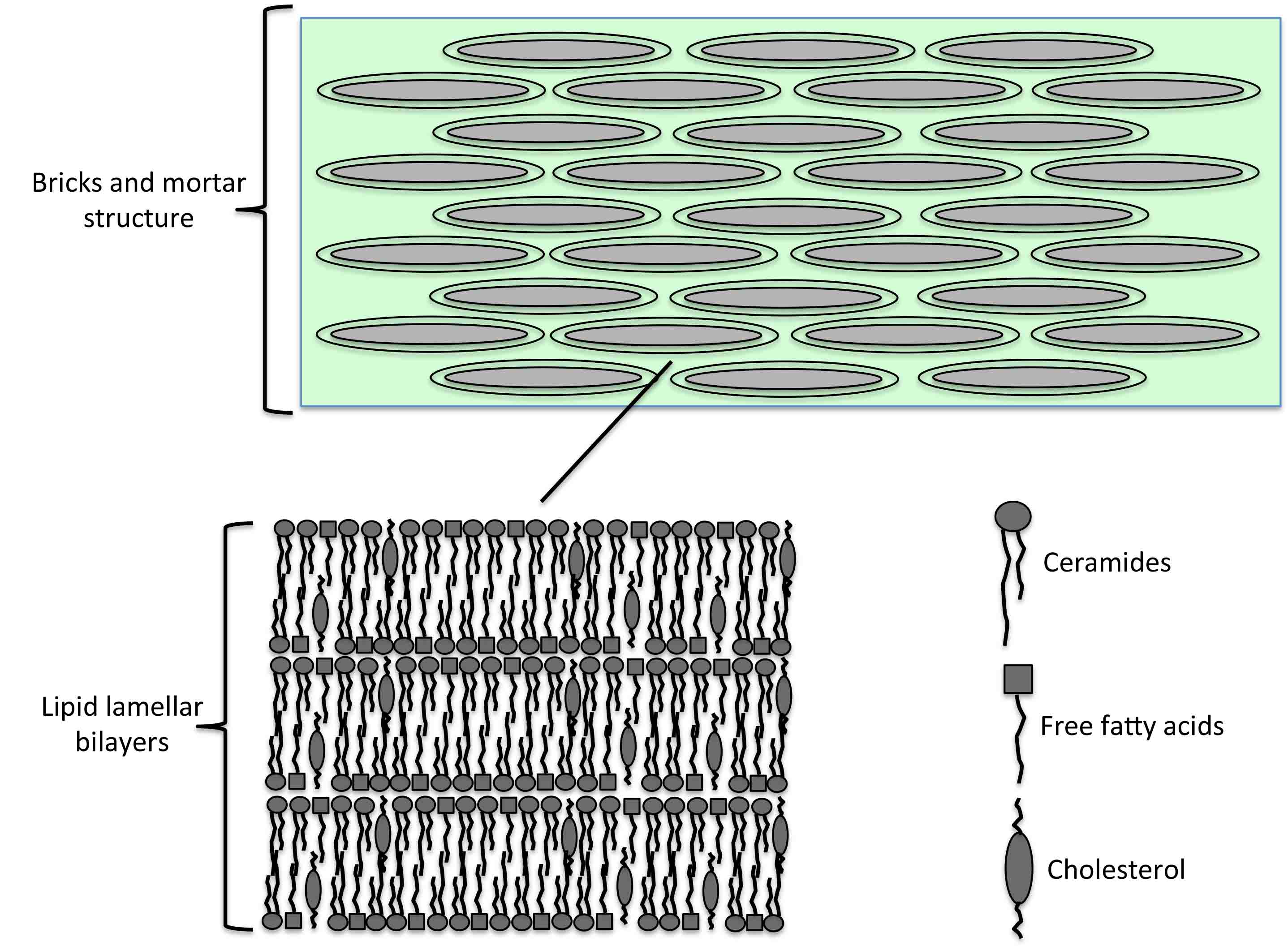
Figure 4. The bricks and mortar structure of the stratum corneum. Corneocytes (bricks) are embedded in a lipid matrix (mortar). The lipid components mostly consist of ceramides, free fatty acids, and cholesterol, which are arranged into lipid lamellar bilayers.
Depending on anatomical location, the stratum corneum can be 15-20 layers of corneocyte cells thick. Corneocytes are flat, polyhedral-shaped cells that contain approximately 80% keratin. The thickness of the stratum corneum depends on sex, age, and disease state. In comparison with basal cell keratinocytes, corneocytes have a much greater surface area; approximately 700 μm2 to 20 μm2.5 This phenomenon is due to the keratinization of keratinocytes, as they become filled with keratin, which aggregates in a planar arrangement.
As already noted, the lipid matrix of the stratum corneum mostly consists of ceramides, free lipids, and cholesterol in equimolar quantities.6-8 In addition, there are small quantities of the following lipids: glucosyl ceramides, cholesterol sulfate, and cholesterol esters.9
For illustration, the molecular structures of three principal lipid classes in skin are shown in Figures 5 and 6. In the plasma membrane of most cells in the body, cholesterol plays an important role in regulating the fluidity of phospholipid bilayers. More than likely, it serves a similar purpose in the stratum corneum. Interestingly, it has been shown that in patients with ichthyoses vulgaris—a dry skin condition, in which skin has a scaly, flaky appearance and cholesterol metabolism is defective—that treatment with cholesterol ameliorates the symptoms.10 The structure of palmitic acid is shown in Figure 5 as an example of one of the free fatty acids, although one should bear in mind that palmitic acid (C16:0) along with stearic acid (C18:0) and oleic acid (C18:1) represent the majority of the free fatty acids in the stratum corneum.
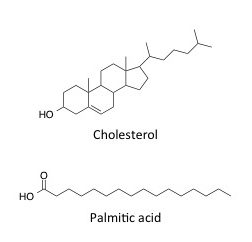
Figure 5. Molecular structures for cholesterol and palmitic acid—lipids found in the stratum corneum lipid matrix.
The ceramides are an important class of lipids, which differ from phospholipids as they contain a less polar head group and more hydrophobic alkyl tail. While the ceramides and phospholipids may appear to form similar membrane type structures, the ceramides clearly form a less permeable barrier in terms of water loss and the entry of foreign particles into the skin. Ceramides consist of three structural units: sphingosine base, amide-linked fatty acid, and possibly an ester group attached to an omega fatty acid. The key provided in Figure 6 allows one to apply the correct nomenclature to the ceramides .11 For example, each ceramide is constructed of one of three possible bases: sphingosine (S), phytosphingosine (P), or 6-hydroxysphingosine (H). Depending on which base is present, its respective letter is utilized as the first letter in naming the ceramide. The amide-linked fatty acid portion of the molecule can include one of three types of fatty acid: normal fatty acid (N), alpha-hydroxyacid (A), or omega-fatty acid (O). Depending which fatty acid is present; its corresponding letter is the second letter in the ceramide name. Finally, in some ceramides an ester group is present and is attached to a pendant omega-fatty acid. In this case the letter E is applied as the last letter of the ceramide name. If there is no ester group present along the upper chain of the ceramide structure, no letter is utilized.
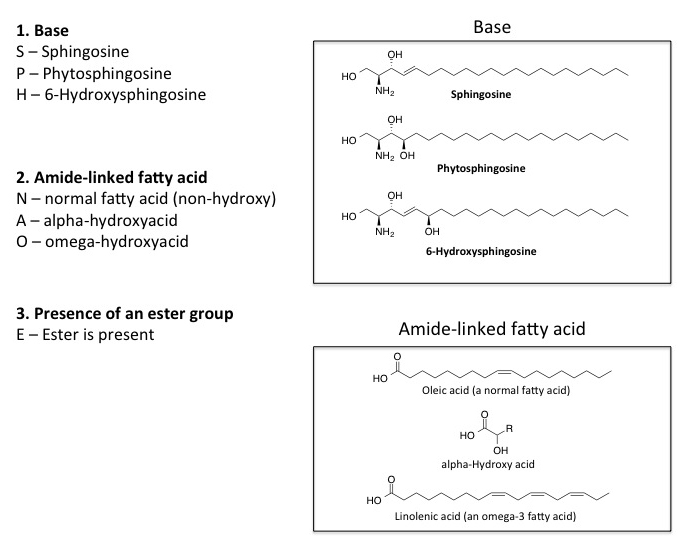
Figure 6. Components of ceramide molecules and their nomenclature.
Figure 7 contains the structures of important ceramides found in skin. Based on the nomenclature scheme already presented, one may deduce the structural components comprising the overall ceramide molecule. One can clearly identify the base—lower portion of the molecule in the structural representation—as well as the type of amide-linked fatty acid that is present. The three types of bases (sphingosine, phytosphingosine, and 6-hydroxysphingosine) can be discerned from top to bottom in both the left and right column and are indicated by their respective symbols, S, P, and H. In addition, the ceramides in the left column contain a normal amide-linked fatty acid (N), while those on the right contain an alpha-hydroxy acid (A). None of the structures shown in Figure 7 contain an ester component.
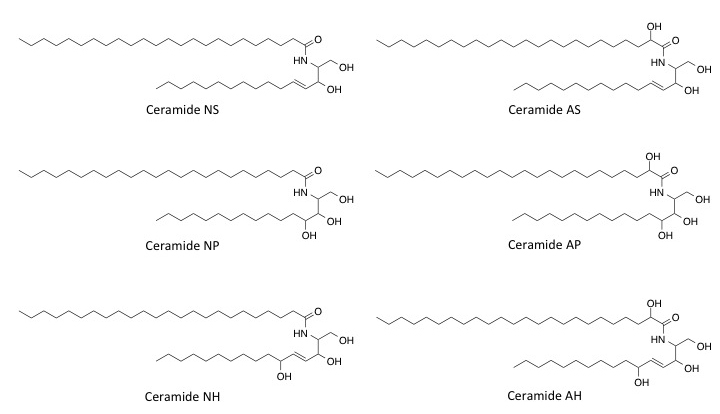
Figure 7. Important ceramides in the stratum corneum.
It is important to note that linoleic (C18:2), linolenic (C18:3), and arachidonic acid (C20:4) also play an important role in the stratum corneum lipid matrix. These are essential omega fatty acids—obtained in the diet—that are often incorporated into ceramide structures. Lignoceric acid (C24:0) is also an important fatty acid that is incorporated into sterol and ceramide structures. Figure 8 includes the structures for Ceramide EOS and Ceramide EOH, two structures that contain an ester functionality along with a pendant omega-6 fatty acid chain.
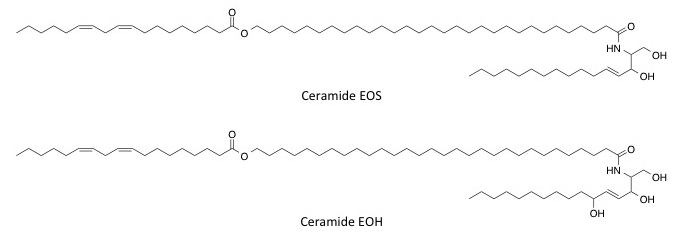
Figure 8. Stratum corneum ceramides containing ester functionality and containing an omega-6 fatty acid chain.
Other important ceramides are those that are covalently attached to corneocytes. In addition, the phase behavior of stratum corneum lipids is a very intriguing topic and certainly relevant to understanding permeation and water loss in the stratum corneum. A considerable amount of evidence suggests that stratum corneum lipids exist in a gel and crystalline phase, thereby providing the skin with an extremely robust barrier function.
References
1. A.-A. Raymond, A. Gonzalez de Peredo, A. Stella, A. Ishida-Yamamoto, D. Bouyssie, G. Serre, B. Monsarrat, and M. Simon, Lamellar bodies of human epidermis: proteomics characterization by high throughput mass spectrometry and possible involvement of CLIP-170 in their trafficking/secretion, Mol. Cell. Proteomics, 7, 2151-2175 (2008).
2. P. Fleckman, B.A. Dale, and K.A. Holbrook, Profilaggrin, a high-molecular-weight precursor of filaggrin in human epidermis and cultured keratinocytes, J. Invest. Dermatol., 85, 507-512 (1985).
3. D. Hohl, T. Mehrel, U. Licht, M.L. Turner, D.R. Roop, and P.M. Steinert, Characterization of human loricrin, J. Biol. Chem., 266, 6626-6636 (1991).
4. M.B. Yaffe, H. Beegen, and R.L. Eckert, Biophysical characterization of involucrin reveals a molecule ideally suited to function as an intermolecular cross-bridge of the keratinocyte cornified envelope, J. Biol. Chem., 267, 12233-12238 (1992).
5. B. Forslind and M. Lindberg, “Structure and function of the skin barrier: an introduction,” in Skin, Hair, and Nails: Structure and Function, Chap. 2, Eds. B. Forslind and M. Lindberg, CRC Press: Boca Raton, FL (2003).
6. P.W. Wertz and D.T. Downing, “Epidermal lipids” in Physiology, Biochemistry, and Molecular Biology of the Skin, Ed. L. Goldsmith, Oxford University Press: New York (1991).
7. N.Y. Schurer and P.M. Elias, The biochemistry and function of stratum corneum lipids, Adv. Lipid Res., 24, 27-56 (1991).
8. A. Weerheim and M. Ponec, Determination of stratum corneum lipid profile by tape stripping in combination with high-performance thin-lay chromatography, Arch. Dermatol. Res., 293, 191-199 (2001).
9. J.A. Bouwstra and G.S. Gooris, The lipid organization in human stratum corneum and model systems, TOJD, 4, 10-13 (2010).
10. T.G. Polefka, “Surfactant interactions with skin” in Handbook of Detergents – Part A: Properties, Ed. G. Broze, Marcel Dekker: New York, NY (1999).
11. S. Motta, M. Monti, S. Sesana, R. Caputo, S. Carelli, and R. Ghidoni, Ceramide composition of the psoriatic scale, Biochim. Biophys. Acta, 1182, 147-151 (1993).
RETURN TO STRUCTURE OF SKIN MENU