

Introduction
Alpha hydroxy acids (also known as AHAs) are small organic acids that consist of a substituent hydroxyl group on the alpha carbon, which is located adjacent to the carboxyl group carbon. Some of the common alpha hydroxy acids are citric acid, glycolic acid, lactic acid, malic acid, and tartaric acid.1 They are widely used in the cosmetics industry and applied topically to treat acne as well as to firm, smooth, and moisturize the skin. Treatment with alpha hydroxy acids also improves skin appearance due to scars, chronological aging, and photo-damage.1,2 Of all the alpha hydroxyl acids, glycolic acid is considered the most useful since it facilitates the shedding of dead skin and helps to even skin discoloration.3 Glycolic acid—also known as hydroxyacetic acid—is the simplest and most widely used of all the alpha hydroxy acids. It can be extracted from plants or prepared synthetically. In nature, glycolic acid is obtained from the sugar of various types of fruit and milk, but mostly comes from sugar cane on an industrial scale.

Figure 1: Molecular structure of glycolic acid.
Mechanism of Action
Glycolic acid is a common cosmetic ingredient, which is known for its properties as an exfoliative agent. It has anti-inflammatory, antioxidant, and keratolytic effects.4 Reportedly, glycolic acid has inhibitory effects on UV-induced skin tumorigenesis in the hairless mouse model.5,6 Unfortunately, only a very limited number of studies demonstrate this property of glycolic acid.
Glycolic acid has the lowest molecular weight of all the alpha hydroxy acids, which renders it with the ability to penetrate skin very easily.4 There have been two proposed mechanisms by which glycolic acid functions. The first theory is that glycolic acid stimulates the epidermis to produce new cells by inducing a mild sub-clinical irritation. The second theory suggests that glycolic acid weakens the intercellular bonding of the corneocytes.7 At low concentration (between 2% and 5%), glycolic acid enhances progressive weakening of cohesion of the intercellular material (corneodesmosomes) of the stratum corneum, which results in a steady exfoliation of the outermost layers (known as the stratum disjunctum) causing desquamation.8
The function of glycolic acid is pH dependent.9,10 It can exist as the free acid (protonated form) or glycolate ion (deprotonated form). In solution, or in formulation, there is an equilibrium that exists between these two species (see Figure 2).
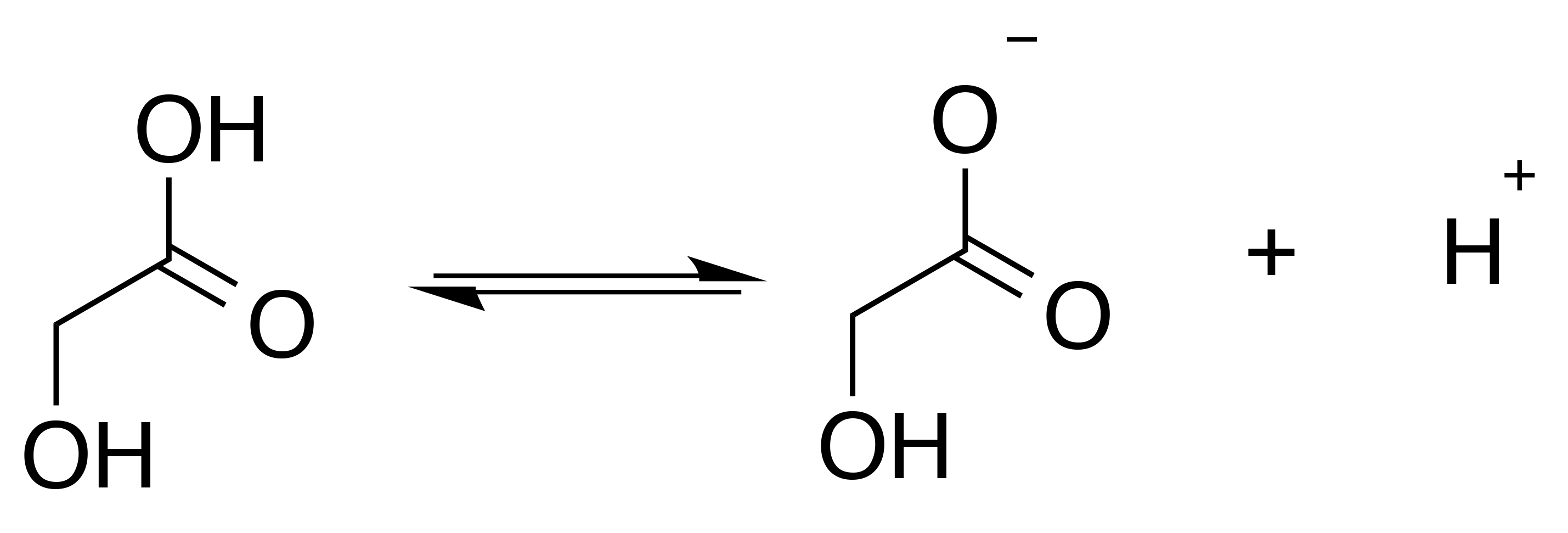
Figure 2: Equilibrium between the glycolic acid free acid and glycolate ion forms.
The free acid form of the molecule (at low pH) is the most biologically active form. In part, this is believed to stem from the ability of the free acid to more freely penetrate the skin. The glycolate ion, which normally would be associated with a weak alkali metal, does not have the ability to penetrate into the skin as freely as the free acid form of the molecule.10 Therefore, the free acid form has a greater degree of bioavailibity.
Treatment of Skin with Glycolic Acid
As already mentioned, glycolic acid is used to treat a number of skin conditions in which it acts as an exfoliating agent. It is most commonly used as the active ingredient in chemical peels to treat a variety of conditions including acne, acne scars, melasma, postinflammatory hyperpigmentation, photoaging, and seborrhea.4 Numerous studies have been carried out with glycolic acid, demonstrating its efficacy in rejuvenating the skin.10
Glycolic acid treatments are most commonly carried out as chemical peels, which entails applying a solution of the alpha hydroxy acid to the skin. Essentially, the skin is wounded, which results in its stimulation during the wounding process. Dead skin tissue is removed during this process. A number of factors effect the degree of exfoliation such as the concentration of the acid, formulation pH, time of contact, and the condition of the skin. Peels can be superficial, medium depth, or deep, which depends on the concentration of glycolic acid.4
Over the last several decades, a number of interesting studies have appeared in the literature with a primary focus on the effects of glycolic acid on the skin. The effects of glycolic acid treatment on dermal matrix metabolism of keratinocytes and fibroblasts were investigated by researchers at Kobe University in Japan.11 Their studies demonstrated that glycolic acid treatment leads to the upregulation of collagen production in fibroblasts. In addition, it was found that cytokines (e.g., IL-1 alpha), released by keratinocytes, play an important role in modulating collagen synthesis and matrix degradation. In skin equivalent models, glycolic acid has been shown to induce an epidermal proliferative response by keratinocyte induction.12
A great deal of interest has been in exploiting glycolic acid treatment for photodamaged skin. Treating sun damaged skin with glycolic acid increases epidermal thickness, hyaluronic acid level of the dermal and epidermal layers, and collagen gene expression, which improves the appearance, texture, and function of the skin.13 Glycolic acid also helps correct skin discoloration by treating pigmentary lesions, which include melasma, solar lentigines, and post-inflammatory hyperpigmentation.4 The suggested hypothesis is that glycolic acid achieves this by remodeling the epidermal layer and accelerating desquamation, which results in enhanced pigment dispersion. Studies have also shown that glycolic acid inhibits melanin formation in melanocytes by minimizing the enzyme activity of tyrosinase without affecting cell growth.14
Glycolic acid has also been shown to have anti-inflammatory properties. To better understand this phenomenon, studies were conducted on the inflammasome complex (NLRP3, NLRC4, AIM2, and ASC ) and revealed that glycolic acid silences the expression of key genes.15 Essentially, the inflammasome complex triggers the inflammatory response. Therefore, by impeding the activity of the inflammasome complex one may reduce inflammation. It was shown that glycolic acid treatment promotes hypermethylation of promoters of key genes (NLRC4 and ASC), which is thought to inhibit the expression and production of inflammasome complex in HaCaT cells (keratonicytes).15 In NHEK (normal human epidermal keratinocytes) cells, reduced levels of interleukin 1 beta—an important modulator of the inflammatory response—are found in glycolic acid treated skin.16
Alpha Hydroxy Acid Use and Exposure to UV Radiation
Even though glycolic acid—one of the most commonly used alpha hydroxyl acids used in skin products—has many useful applications, there have been concerns for its use, especially higher concentrations. For example, subjects treated with 10% glycolic acid became sensitized to UV light as measured by an increase in the number of sunburn cells and a decrease in minimal erythema dose (MED).17 It should be noted that this effect is reversible after stopping treatment for 1 week. Further studies from the same group compared sensisitivity to solar radiation of glycolic acid and salicylic acid (a beta hydroxy acid).18 Their results demonstrated that treatment with glycolic acid prior to exposure to solar radiation resulted in adverse reactions of the skin while treatment with salicylic acid did not.
There have been further concerns about the use of high concentrations of glycolic acid as chemical peels in the news media, although they did not cite any published research studies.19 Regardless, they address important labeling concerns, which could provide consumers with a cautionary statement that indicates that alpha hydroxy acids may burn the skin or cause inflammation or pain.
In any event, it should be pointed out that a comprehensive study conducted by the National Institutes of Health in the United States—part of the National Toxicology Program—found that salicylic acid had no effect on UV-induced carcinogenesis.20 Tests were carried on the hairless mouse in which the mice were treated with salicylic acid (concentrations of 4% and 10%) and exposed to simulated solar radiation 5 days per week for 40 weeks.
Are There Alternatives?
Even though the Food and Drug Administration (FDA) has recognized that alpha hydroxy acids have possible harmful effects, they are not banned from use in cosmetic products in the United States. The FDA has suggested that cosmetic companies place warning labels that state the product contains active alpha hydroxyl acid which may increase the skin’s sensitivity to sunburn.19
Polyhydroxy acids (PHAs) and polyhydroxy bionic acids (PHBAs) are novel analogs of alpha hydroxy acids, which have similar anti-aging and skin smoothing properties to alpha hydroxy acids, but with less irritation. The molecular structure of PHAs and PHBAs are similar to alpha hydroxy acids.2
Other active ingredients have been suggested as milder replacements for alpha hydroxy acids while still providing the beneficial effects of enhanced exfoliation of the skin. For example, researchers discovered enzymes in salmon eggs that break down the egg shell, but do not destroy the embryo. These enzymes (proteases) are collected from the fish hatchery water after the larvae are hatched—not harming the fish in the process. It is purported that such enzymes, when applied to skin, act on the corneodesmosines of the stratum corneum thereby facilitating exfoliation. Two of the enzymes—designated as Zonase and Aquabeutine XLTM—have been promoted for use in skin care; however, at this time it is difficult to ascertain if these ingredients are acceptable substitutes for alpha hydroxy acids.21
Concluding Remarks
Alpha hydroxy acids have been widely used as an active cosmetic topical agent for a long time and are derived from natural products including fruit, milk, and sugar cane, which make it easily accessible. Alpha hydroxy acids have proven efficacy to treat many skin conditions such as dry skin, ichthyosis, melasma, wrinkling, acne scars, and blemishes. However, there are concerns about side effects of alpha hydroxy acids. Particular attention has been given to photosensitization of glycolic acid treated skin. The adverse reactions of glycolic acid have led to investigations of alternative therapies such as PHAs, PHBAs, and enzymes that increase the rate of exfoliation in skin. Nevertheless, the responsible use of alpha hydroxy acids at controlled concentrations (less than 10%) as approved by FDA and making the consumer aware of the side effects will ensure safe application of this molecule.
References
1. Alpha Hydroxy Acids. U.S. Department of Health and Human Services. U.S. Food and Drug Administration (2015).
2. A. Kornhauser, S.G. Coelho, and V.J. Hearing, Applications of hydroxy acids: classification, mechanisms, and photoactivity, Clin. Cosmet. Investig. Dermatol., 3, 135–142 (2010).
3. M. Hall, “What products truly promote anti-aging?” Skin Cancer Foundation, www.skincancer.org (2013).
4. J. Sharad, Glycolic acid peel therapy—a current review, Clin. Cosmet. Investig. Dermatol., 6, 281–288 (2013).
5. K.S. Ahn, K.S. Park, K.M. Jung, H.K. Jung, S.H. Lee, S.Y. Chung, K.H. Yang, Y.P. Yun, H.B. Pyo, Y.K. Park, Y.W. Yun, D.J. Kim, S.M. Park, and J.T. Hong, Inhibitory effect of glycolic acid on ultraviolet B-induced c-fos expression, AP-1 activation and p53-p21 response in a human keratinocyte cell line, Cancer Lett., 186, 125-135 (2002).
6. J.T. Hong, E.J. Kim, K.S. Ahn, K.M. Jung, Y.P. Yun, Y.K. Park, and S.H. Lee, Inhibitory effect of glycolic acid on ultraviolet-induced skin tumorigenesis in SKH-1 hairless mice and its mechanism of action, Mol Carcinog., 31, 152-160 (2001).
7. S. Gupta, Hydroxy acids based delivery systems for skin resurfacing and anti-aging compositions, U.S. Patent US20040092482 A1 (2004).
8. M. Fartasch, J. Teal, and G.K. Menon, Mode of action of glycolic acid on human stratum corneum: ultrastructural and functional evaluation of the epidermal barrier, Arch. Dermatol. Res., 289, 404-409 (1997).
9. L.E. Samuels, Understanding the science behind pH and its role in younger, healthier-looking skin, Cosmet. Dermatol., 23, 562-567 (2010).
10. R. Moy, D. Luftman, and L.S. Kakita, Glycolic Acid Peels, Marcel Dekker: New York, NY (2002).
11. Y. Okano, Y. Abe, H. Masaki, U. Santhanam, M. Ichihashi, and Y. Funasaka, Biological effects of glycolic acid on dermal matrix metabolism mediated by dermal fibroblasts and epidermal keratinocytes, Exp. Dermatol., 12 Suppl. 2, 57-63 (2003).
12. S. Denda, M. Denda, K. Inoue, T. Hibino, Glycolic acid induces keratinocyte proliferation in a skin equivalent model via TRPV1 activation, J. Dermatol. Sci., 57, 108-113 (2010).
13. E.F. Bernstein, J. Lee, D.B. Brown, R. Yu, and E. Van Scott, Glycolic acid treatment increases type I collagen mRNA and hyaluronic acid content of human skin, Dermatol. Surg., 27, 429-433 (2001).
14. A. Usuki, A. Ohashi, H. Sato, Y. Ochiai, M. Ichihashi, and Y. Funasaka, The inhibitory effect of glycolic acid and lactic acid on melanin synthesis in melanoma cells, Exp. Dermatol., 12 Suppl. 2, 43-50 (2003).
15. S.C. Tang, J.I. Yeh, S.J. Hung, Y.P. Hsiao, F.T. Liu, and J.H. Yang, Glycolic acid silences inflammasome complex genes, NLRC4 and ASC, by inducing DNA methylation in HaCaT cells, DNA Cell Biol., 35, 124-134 (2016).
16. S.J. Hung, S.C. Tang, P.Y. Liao, J.S. Ge, Y.P. Hsiao, and J.H. Yang, Photoprotective potential of glycolic acid by reducing NLRC4 and AIM2 inflammasome complex proteins in UVB radiation-induced normal human epidermal keratinocytes and mice, DNA Cell Biol., 36, 177-187 (2017).
17. K. Kaidbey, B. Sutherland, P. Bennett, W.G. Wamer, C. Barton, D. Dennis, and A. Kornhauser, Topical glycolic acid enhances photodamage by ultraviolet light, Photodermatol. Photoimmunol. Photomed., 19, 21-27 (2003).
18. A. Kornhauser, R.-R. Wei, Y. Yamaguchi, S.G. Coelho, K. Kaidbey, C. Barton, K. Takahashi, J.Z. Beer, S.A. Miller, and V.J. Hearing, The effects of topically applied glycolic acid and salicylic acid on ultraviolet radiation-induced erythema, DNA damage and sunburn cell formation in human skin, J. Dermatol. Sci., 55, 10-17 (2009).
19. J.E. Allen, A hidden danger in skin care, Los Angeles Times, December 16, 2002.
20. D.A. Casciano et al., Photocarcinogenesis study of glycolic acid and salicylic acid (CAS Nos. 79-14-1 and 69-72-7) in SKH-1 mice (simulated solar light and topical application study), Natl. Toxicol. Program Tech. Rep. Ser., 524, 1-242 (2007).
21. M. Anthonavage, S. Jindal, A. Lindberg, and F. Coste, Aquabeautine XL, a novel, marine-derived, self-limiting enzyme providing effective, gentle exfoliation and clinically demonstrated anti-aging benefits, J. Invest. Dermatol., Research and Therapeutics, 132, S85-S96 (2012).