

By Gopinathan K. Menon
October 15, 2017
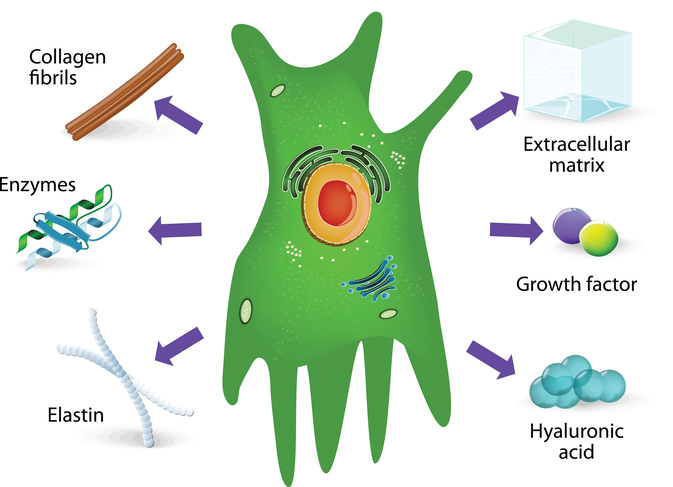
Why Should You Be Interested in this Cell Type?
The quality of dermis, foundation of resilience, and healthy appearance of skin depend primarily on this cell type. It is also one of the most widely used cell culture models for testing cosmetic ingredients—be it for toxicology or efficacy evaluation. It is also used for creating the 3-D models of skin, increasingly being employed as an alternative to animal testing.
What are Fibroblasts and What Do They Do?
As mentioned above, fibroblasts are cells of the dermis (but are also found elsewhere in the body) that produce the dermal matrix composed of collagen, elastin, hyaluronic acid, and glycoproteins. In addition, they also synthesize and secrete several matrix metalloproteinases (MMPs), such as collagenase, gelatinase, and elastase that degrade the dermal matrix, as well as various growth factors and cytokines. In healthy skin, a balance between the production of structural proteins and MMPs assure an orderly remodeling of the dermis. Cytokines are involved in modulating inflammatory responses and cell-to-cell communications (as in dermo-epidermal interactions). As much as these cells influence the functions of other cell types of skin, fibroblast functions, in turn, are also modulated by keratinocytes (and also other cell types) during development, photoaging, and wound healing.1
It is well known that keratinocytes stimulate fibroblasts to synthesize growth factors, which in turn induce proliferation in a paracrine manner. During early development of skin, under the influence of developing epidermis, fibroblasts form localized condensations that in turn induces the focally overlying epidermis to form placodes that differentiate into hair follicles. In later life, in response to signals from UV-damaged epidermis (and also direct effects of radiation), fibroblasts increasingly secrete MMPs that degrade the dermal matrix, while producing less dermal matrix components—the sum total leads to lines and wrinkles that characteristic of photoaging. It is also evident that melanocytes respond to fibroblast derived factors independent of keratinocytes, which could contribute to abnormal pigmentation of photoaged skin as well.2,3 Fibroblasts are also absolutely necessary for healing of skin wounds, as we all know well.
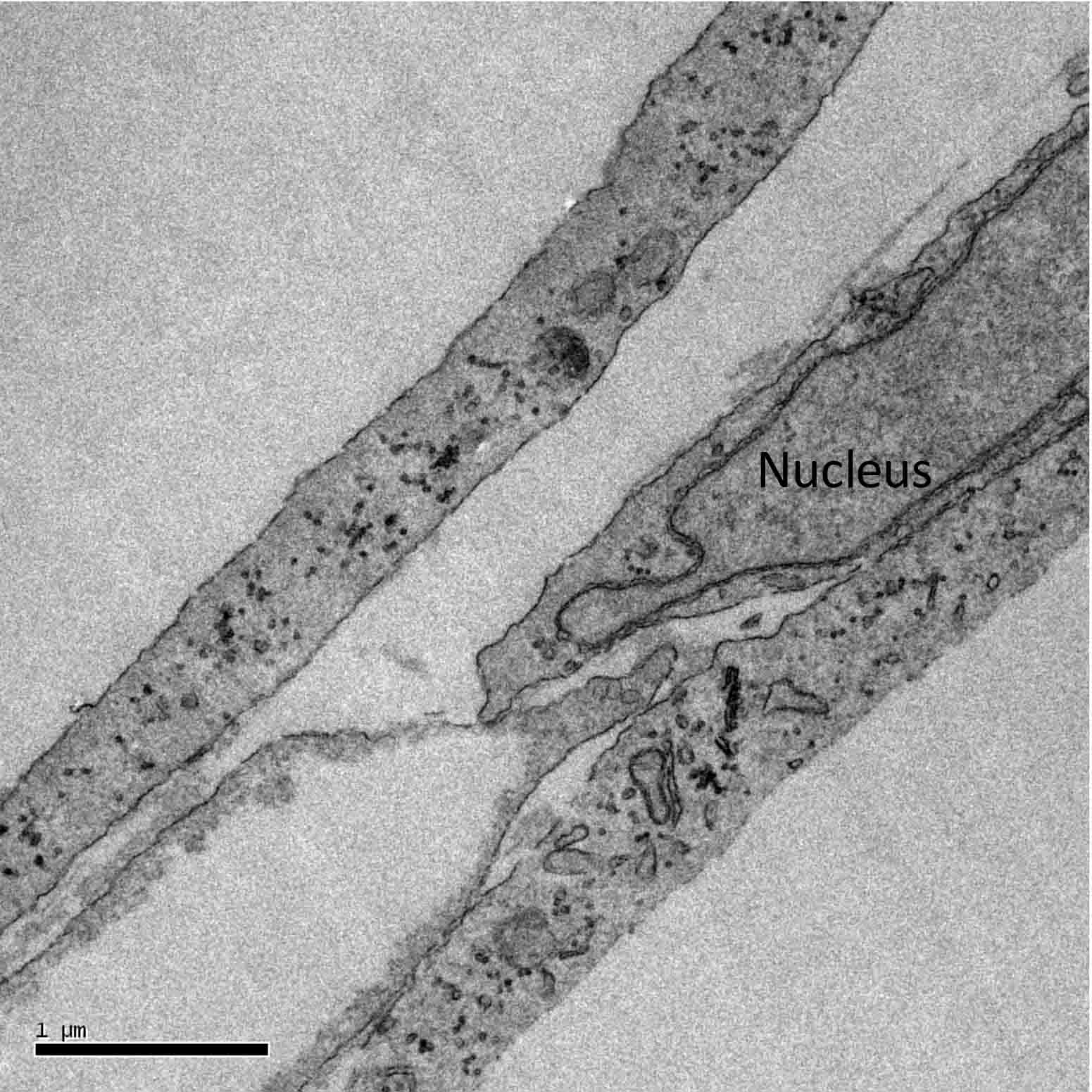
Figure 1: Electron microscope image of fibroblasts in cell culture.
Morphology of the Fibroblast
Fibroblasts are often described as spindle-shaped, which is probably due to their appearance in a culture dish (Figure 1). In vivo, within the dermis, they are definitely stretched out, almost trapped within the fibers of collagen that they secrete (Figure 2), and appear stellate rather than spindle shaped.
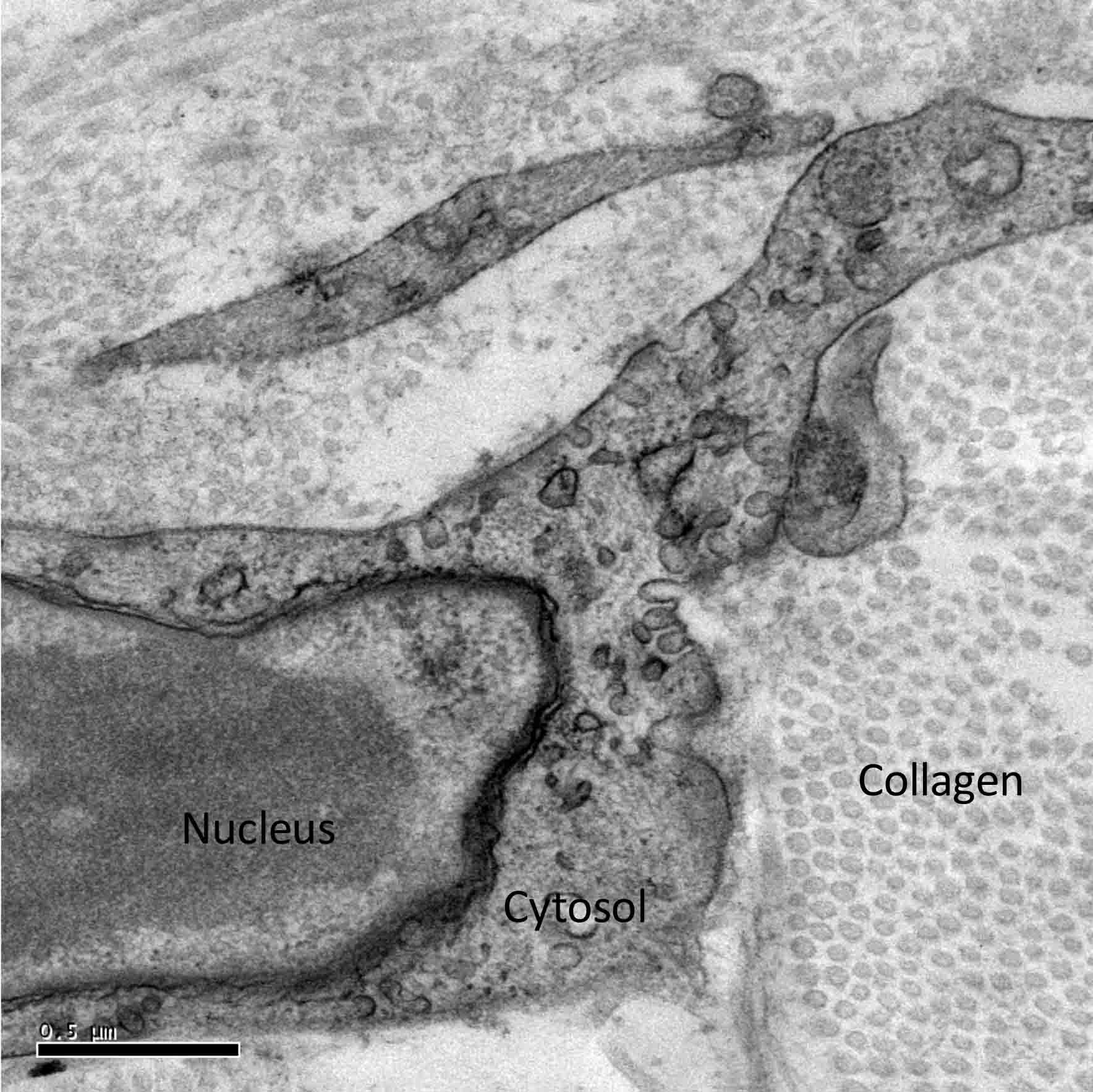
Figure 2: Electron microscope image of a fibroblast in a section of skin.
In the electron microscope, their subcellular appearance reflects their active or inactive status. Fibroblasts that are actively secreting collagen or other matrix components may exhibit varying degrees of development of endoplasmic reticulum, mitochondria, or Golgi complex. This is especially striking in cell cultures (Figure 3), and hence a valuable parameter in evaluating effects of specific ingredients in vitro.
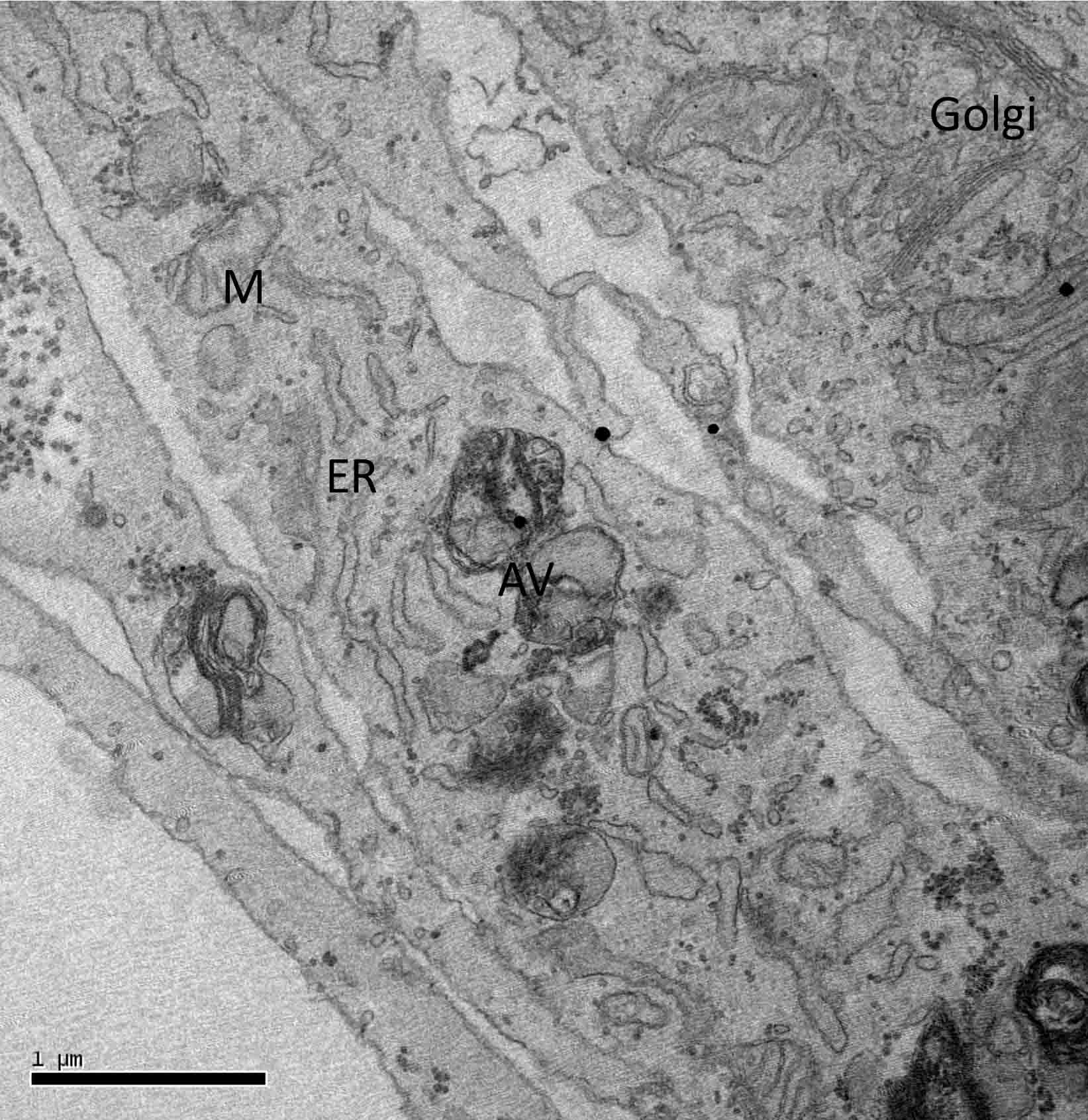
Figure 3: Fibroblasts in culture that have been treated with a cosmetic ingredient. Note the high degree of development of Golgi complex in the cell to the right. Other cells show endoplasmic reticula and autophagic vesicles.This electron microscope image also documents the heterogeneous nature of fibroblasts in the same culture: M=mitochondria, ER=endoplasmic reticula, AV=autophagic vacuoles, and Golgi=Golgi apparatus.
Are all Fibroblasts Created Equal?
As it turns out, they are not. Nearly four decades ago, Harper and Grove showed that fibroblasts from papillary (superficial) dermis from the same skin specimen exhibited greater proliferative capacity than those from the reticular dermis in vitro.4 Later studies showed that normal adult human skin contains at least three distinct subpopulations of fibroblasts, from unique niches in the dermis—papillary dermis, reticular dermis, hair follicles—which showed clear distinctions when cultured separately, and also exhibited differences in their physiology as well.5 Moreover, fibroblasts can acquire a different phenotype called a myofibroblast—expressing the smooth muscle actin—under the control of keratinocytes and a finely tuned balance between a proinflammatory or a transforming growth factor (e.g., TGF-Beta) dominant microenvironment, especially during wound healing. It has been reported that in the early stages of differentiation, fibroblasts show an increase in focal adhesion proteins denoting a proto-myofibroblast phenotype which emerges with mechanical stress on the cells , and further stimulation leads to differentiation to the myofibroblast phenotype. In vitro, it was reported that fibroblasts retain fibroblast markers only in early passage cultures, but with increased cell passages they begin to trans-differentiate into myofibroblasts—with myofibroblast markers (paxillin, vincullin, and tensin) appearing as early as the second passage.6 An implication of this finding is that unless studies are conducted on cells prior to the third passage in culture, results may be clouded by having a highly heterogeneous population of cells, and may not represent a primary fibroblast response to an ingredient under investigation.
Fibroblasts in Senescence
In 1995, Dimri et al. identified a marker (β-glucosidase) that is histochemically detectable at pH 6 in cultures of senescent fibroblasts.7 This marker is expressed in senescent, but not pre-senescent or quiescent fibroblasts. Since then, this marker has been extensively used in experimental cell culture studies for induced senescence as well as in tests designed to reduce or negate the effects of senescence.8 Senescent fibroblasts are also known to create a pro-oncogenic environment that demonstrates synergy with mutations, driving initiation and progression of cancers such as melanoma, suggesting that senescent fibroblasts may be a potential therapeutic target.9
Remodelling of a Collagen Matrix
A cell-free collagen matrix is often used in production of bioengineered 3-D models of skin for skin grafts, in experimental studies, or as an alternate for animal models in cosmetic or pharmaceutical testing of active compounds (see Reference 10). The matrix is seeded with fibroblasts and keratinocytes are subsequently grown on top of the dermal matrix. When fibroblasts are seeded onto a collagen matrix, they remodel the matrix, causing contraction of the gel. This process involves an overall morphology change in fibroblasts from a dendritic to stellate/bipolar structure, and cell–matrix interactions mature from punctate to focal adhesion organization.11
Role of Fibroblasts in Scarring and Scar-less Wound Healing
Wound healing depends on synthesis and/or remodeling of the extracellular matrix by fibroblasts. In addition to resident fibroblasts at the wound site, other fibroblasts are also recruited to the site of the wound by chemokines an cytokines released by macrophages and neutrophils. Myofibroblasts appear in the adult wound about a week after injury. The content of myofibroblasts is greatest during the second and third week and then decreases with time. They synthesize extracellular matrix components, such as collagen types I and III, and with re-epitehlialization of the wound, undergo apoptosis.12 The number of myofibroblasts correlates with the wound contraction and the degree of scarring.13
Fetal wounds heal without scars—they have less inflammatory cells and cytokine expression, yet heal more rapidly than adult wounds. This is explained by intrinsic differences between adult and fetal fibroblasts in synthesis of extracellular matrix. In vitro, fetal fibroblasts synthesize more type III and IV collagen than fibroblasts from adults. While fetal fibroblasts synthesize collagen when they proliferate, adult fibroblasts have delayed collagen synthesis during proliferation. Fetal fibroblasts possess more surface receptors for hyaluronic acid, which aids in enhanced fibroblast migration. Further, TGF-Beta is decreased in the fetal wound. Thus, the fetal fibroblasts have an innate ability to synthesize extracellular matrix and generate an organized dermis at wound sites.13
Differences in contractile fibroblasts (i.e. myofibroblasts) have also been reported. Myofibroblasts, detected by the presence of alpha-smooth muscle actin, appear in the adult wound one week after wounding. The content of myofibroblasts is greatest during the second and third week and then decreases with time, whereas wounds made early in gestation have virtually no myofibroblasts.14 In contrast, scarring fetal and postnatal wounds have progressively more active myofibroblasts, which correlates with contraction and degree of scarring.15 Overall, the fetal fibroblast has the intrinsic ability to synthesize a dermal extracellular matrix that is superior to that produced by the adult fibroblast in terms of its ability to generate an organized dermis at sites of injury. On the other hand, there are fibrotic diseases such as scleroderma and keloid scars, which result from excessive accumulation of collagen in the extracellular matrix due to the continued proliferation/ activation of myofibroblasts. As the fibrocontractive diseases have implications beyond skin, extending to the heart, lungs, and kidney, there is a lot of research interest in the physiology and pathology of myofibroblasts.6 Nearly 45% of all deaths in the developed world are attributed to some type of chronic fibroproliferative disease, which attests to the significance of fibroblasts to the biomedical sciences.16
References
1.
S. Werner, T. Krieg, and H. Smola, Keratinocyte-fibroblast interactions in wound healing, J. Invest. Dermatol., 127, 998-1008 (2007).
2. P. Sirimahachaiyakul, R.F. Sood, L.A. Muffley, M. Seaton, C.T. Lin, L. Qiao, J.S. Armaly, A.M. Hocking, and N.S. Gibran,
Race does not predict melanocyte heterogeneous responses to dermal fibroblast-derived mediators, PLoS One, 10(9), e0139135 (2015).
3. X.H. Yuan and Z.H. Jin, Paracrine regulation of melanogenesis, Br. J. Dermatol., doi: 10.1111/bjd.15651, epub (2017).
4. R.A. Harper and G. Grove,
Human skin fibroblasts derived from papillary and reticular dermis: differences in growth potential in vitro, Science, 204, 526-527 (1979).
5. J.M. Sorrell and A.I. Caplan, Fibroblast heterogeneity: more than skin deep, J. Cell. Sci., 117, 667-675 (2004).
6. J. Baum and H.S. Duffy, Fibroblasts and myofibroblasts: what are we talking about? J. Cardiovasc. Pharmacol., 57, 376-379 (2011).
7. G.P. Dimri, X. Lee, G. Basile, M. Acosta, G. Scott, C. Roskelley, E.E. Medrano, M. Linskens, I. Rubelj, and O. Pereira-Smith,
A biomarker that identifies senescent human cells in culture and in aging skin in vivo, Proc. Natl. Acad. Sci. USA, 92, 9363-9367 (1995).
8. A. Lebleu, A. Fuet, G. Menon, J.M. Botto, C. DalFarra, and N. Domloge,
FOXO3a transcription factor is implicated in fibroblasts and skin senescence, J. Invest. Dermatol., 131(Suppl.1), S50 (2011).
9. E. Kim, V. Rebecca, I.V. Fedorenko, J.L. Messina, R. Mathew, S.S. Maria-Engler, D. Basanta, K.S.M. Smalley, and A.R.A. Anderson, Senescent fibroblasts in melanoma initiation and progression: an integrated theoretical, experimental, and clinical approach, Cancer Res., 73, 6874-6885 (2013).
10. I. Jones, L. Currie, and R. Martin, A guide to biological skin substitutes, Br. J. Plast. Surg., 55, 185-193 (2002).
11. E. Tamariz and F. Grinnell,
Modulation of fibroblast morphology and adhesion during collagen matrix remodeling, Mol. Biol. Cell., 13, 3915-3929 (2002).
12. A. Desmouliere, M. Redard, I. Darby, and G. Gabbiani, Apoptosis mediates the decrease in cellularity during the transition between granulation tissue and scar, Am. J. Pathol., 146, 56-66 (1995).
13. B.J. Larson, M.T. Longaker, and H.P. Lorenz,
Scarless fetal wound healing: a basic science review, Plast. Reconstr. Surg., 126, 1172-1180 (2010).
14. R.A.F. Clark, Wound Repair Overview and General Considerations, Vol. 12, Plenum Press: New York, 1996.
15.
J.M. Estes, J.S. Vande Berg, N.S. Adzick, T.E. MacGillivray, A. Desmoulière, and G. Gabbiani, Phenotypic and functional features of myofibroblasts in sheep fetal wounds, Differentiation, 56, 173-181 (1994).
16. T.A. Wynn, Cellular and molecular mechanisms of fibrosis, J. Pathol., 214, 199-210 (2008).