

By Carine Mainzer
June 15, 2018
LCs are immune sentinels located in the epidermal layer of skin. With their long dendrites, which protrude through keratinocytes tight junctions to capture external antigens, they are able to sense danger at the surface of the skin and trigger, accordingly, inflammatory or tolerant immune responses.1 LCs are key players in inflammatory skin diseases including irritant and allergen-induced contact dermatitis, and are often used to assess the safety of personal care products.2,3
LCs are a specific type of antigen presenting cells (APCs) located in the suprabasal layers of the epidermis and represent 3-5% of epidermal cells under homeostatic conditions.4 Their role is to sense the external environment for pathogens and to alert the immune system of danger.
Because of the features they share with dendritic cells (shape, morphology, ability to migrate to the lymph nodes), and the fact that under inflammatory conditions, LCs can be replenished from blood-derived monocytes, LCs have been thought for a long time to originate from myeloid progenitors. However, this paradigm is changing as increasing evidence demonstrates that LCs originate during embryonic development from macrophage progenitors that emerge directly from the yolk sac or from the fetal liver after differentiating into fetal monocytes (Figure 1). These progenitors then seed the skin and LCs acquire their full phenotype shortly after birth through local epidermal signals.5,6 In addition to these findings, LCs are able to self-renew under steady state conditions independently of blood monocytes.5
Since LCs gain their full identity in a short timeframe after birth, it is necessary to investigate whether additional factors, such as the establishment of the microbiome, either directly or indirectly contribute to the local differentiation of LCs.
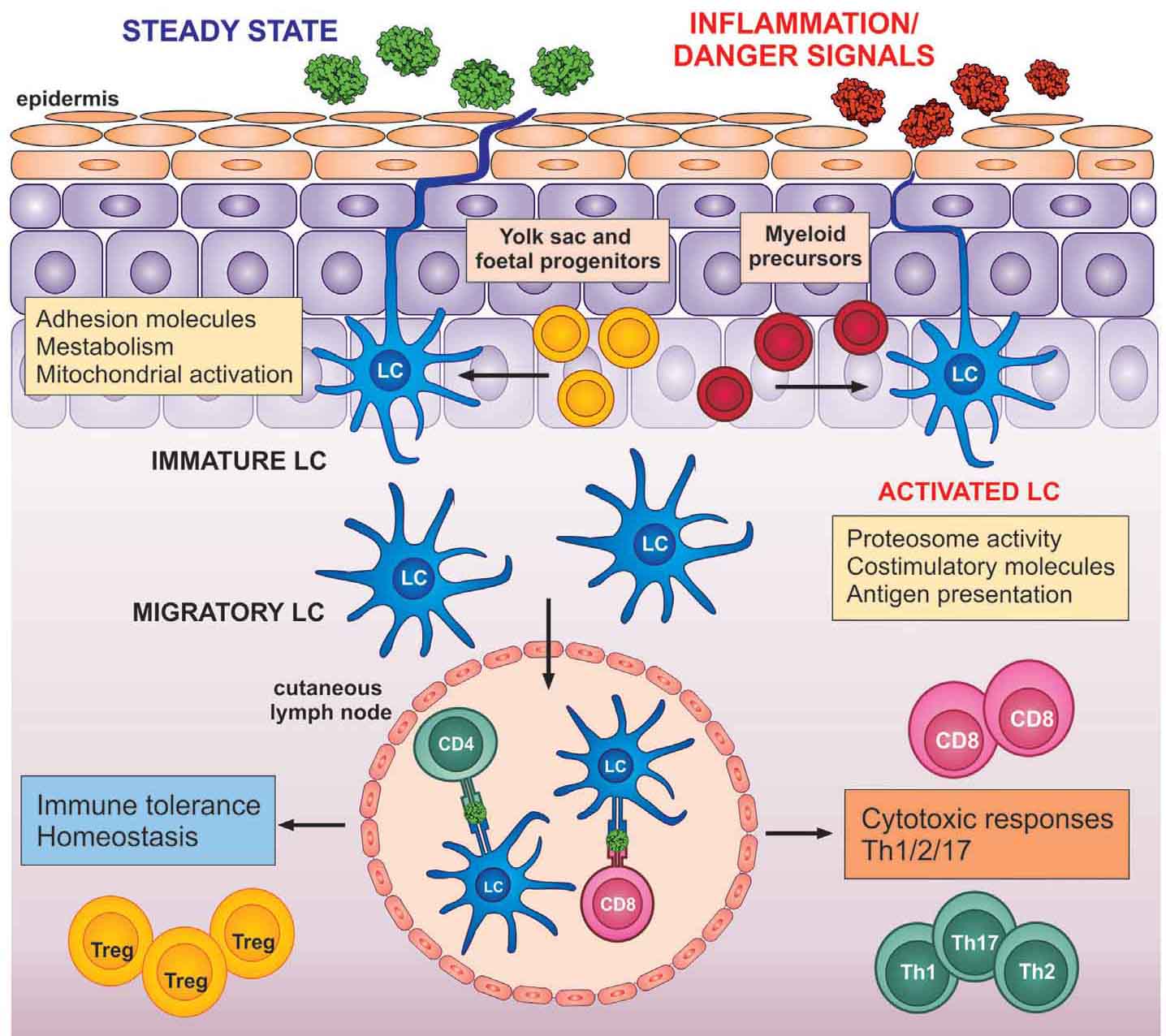
Figure 1: Regulation of the adaptive immune system by LCs under steady state or inflammatory conditions. LCs are largely derived from embryonic fetal liver monocytes and yolk-sac derived macrophages (yellow), but can be replenished by myeloid precursors (red) under high inflammation. In the steady state, LCs express adhesion molecules that play a role in metabolism and activation of mitochondria, whereas in the activated state, LCs are less adhesive, display higher proteasome activity, and express molecules involved in T cell activation (costimulatory and antigen presenting molecules). Both steady state and activated LCs can migrate to local lymph nodes and regulate the response of the adaptive immune system (tolerance vs. cytotoxic responses). Illustration reprinted under the Creative Commons Attribution License (CC BY) from K. Clayton, Langerhans cells—programmed by the epidermis, Front. Immunol., 8, 1676 (2017); doi: 10.3389/ mmu.2017.01676. Copyright ( 2017), Clayton, Vallejo, Davies, Sirvent, and Polak.
LCs have a dendritic shape and are characterized by the presence of electron dense small racquet shaped organelles called Birbeck granules in their cytoplasm (see Figure 2).7,8 The function of Birbeck granules is still unclear, although a role in pathogen retention and degradation has been demonstrated.9 Under homeostatic conditions, LCs express characteristic surface markers. Some of the most noteworthy ones are:
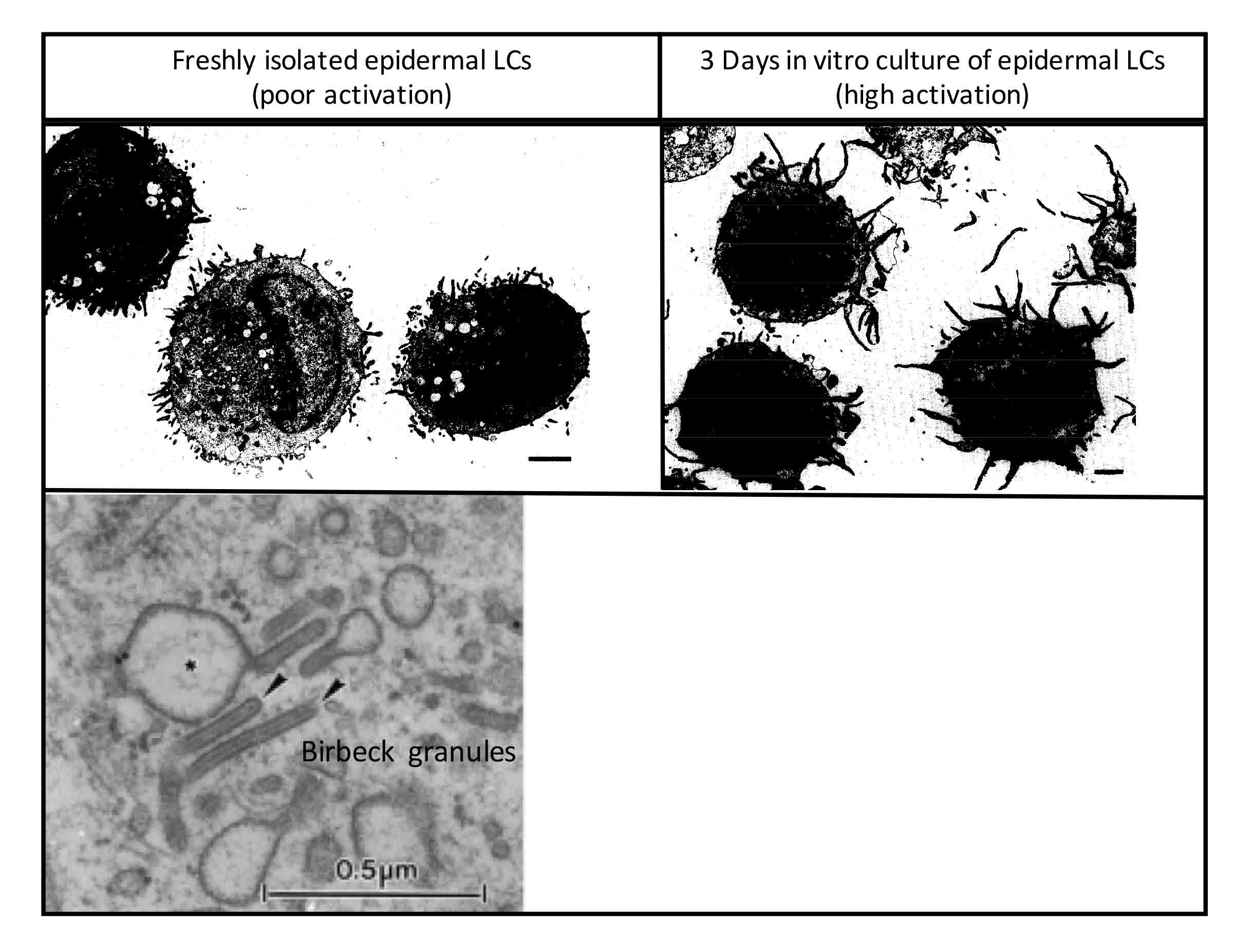
Figure 2: Specific morphology and structures of epidermal Langerhans cells. The two binary micrographs (above) reprinted from Journal of Investigative Dermatology, Vol. 94, M.B.M. Teunissen et al., Human epidermal Langerhans cells undergo profound morphologic and phenotypical changes during in vitro culture, pp. 166-173, Copyright (1990), with permission from Elsevier. The transmission electron micrograph (below) reprinted from Journal of Investigative Dermatology, Vol. 120, D. Lipsker et al., Cored tubules are present in human epidermal Langerhans cells, pp. 407-410, Copyright (2003), with permission from Elsevier.
LCs have the unique capacity of self-renewal and it is estimated that about 2-3% of LCs actively cycle in the human epidermis in the steady state (homeostatic conditions).15 Their development and maintenance requires several factors including transforming growth factor-β1 (TGF-β1), interleukin-34 (IL-34), colony stimulating factor-1 (CSF-1), and bone morphogenetic protein-7 (BMP-7).
TGF-β1 and transcription factors, and members of the TGF-β1 signaling network such as runt-related transcription factor 3 (Runx3), inhibitor of DNA binding 2 (Id2), PU.1, and P14, are required for the establishment of LCs.5 TGF-β1 is secreted by LCs in an autocrine manner and its activation is mediated through keratinocyte-derived integrins—αvβ6 and αvβ8. TGF-β1 is important to maintain epidermal residence of LCs.4
BMP-7 is a member of the TGF-β superfamily that is expressed in the epidermis. Its link with the differentiation of LCs has been recently highlighted as newborn mice lacking BMP7 were observed to have reduced levels of LCs that were morphologically immature.16
CSF-1R and its ligands, CSF-1 or IL-34, have been shown to be involved in the maintenance of LCs, as mice lacking CSF1R have lower levels of LCs.17
During inflamed conditions, when epidermal LCs emigrate from the epidermis, replenishment is mediated by circulating monocytes and is dependent on CCR2 and CCR6 expression and macrophage colony-stimulating factor (M-CSFR) signaling.15 As an illustration, it has been shown that circulating monocytes can differentiate intro CCR6+ langerin+ cells in vitro in response to TGF-β.
LCs sense the external environment constantly by extending their dendrites through epidermal tight junctions to capture foreign antigens (any molecules from pathogens such as bacteria, viruses, fungi, and parasites, or haptens, i.e. any small molecule able to trigger immune responses, such as chemicals). During this process, LCs form de novo tight junctions with keratinocytes to maintain barrier integrity.1
LC activation occurs through antigen recognition and inflammatory signals. Antigen recognition is mediated through various surface receptors including pattern recognition receptors (PRRs), such as the toll-like receptors (TLRs). Each dendritic cell has a defined set of TLRs allowing it to adapt to the variety of pathogens it can recognize. Human LCs are equipped with a limited set of TLRs involved in the recognition of bacteria and viruses: TLRs 1, 2 (low), 3, 5, 6, 7, 8, and 10 (low).18,19 However, LCs do not efficiently internalize bacteria in vitro, leading to the suggestion that they preferentially prime antiviral immunity.20,21
The release of inflammatory cytokines, especially of the IL-1 family and tumor necrosis factor-α (TNF-α) by activated keratinocytes, is also an important factor that will complete LC activation and induce structural and molecular changes.22 The morphological changes occurring in epidermal LCs are necessary for their mobility and their role of priming T cells. During this process, LCs downregulate molecules retaining them in the epidermis (E-cadherin, CCR6) and upregulate molecules involved in their migration (epithelial cell adhesion molecule (EpCAM), CCR7—chemokine receptor homing LCs in the lymph nodes). LCs upregulate MHC-II, extend their dendrites, express co-stimulatory molecules (CD80, CD86, CD83) and secrete specific cytokines, all signals involved in priming naive T cells in the lymph nodes.15 LCs induce the differentiation of naive T cells into different subsets of T cells depending on the antigen recognized and the cues from the microenvironment.
Human LCs have been shown to induce the differentiation of naive CD4+ T cells into T helper cells (Th1, Th2, Th17, Th22)23-25 and regulatory T cells (Treg) (see Figure 3).26 Plus, LCs have been shown to be powerful inducers of CD8+ T cells through cross presentation (presentation of extracellular proteins on MHC-I) and production of IL-15.22,27 It is important to underline that most of these findings come from in vitro experiments. The few experiments realized in vivo, seem to highlight a milder involvement of LCs in priming naive T cells into different Th populations with a more prominent role for dermal dendritic cell subsets.4 In fact, human skin LCs seem to have a reduced ability to take up bacterial antigens, due to lower levels of CD32 (one receptor enabling phagocytosis) and deficiency in processing or presenting restricted antigens on MHC-II receptors,20 which makes them more prone to activate Treg. Interestingly, human epidermal LCs have been shown to activate and induce proliferation of skin resident Treg under homeostatic conditions.26 It was proposed that these LCs could present self-antigens or possibly antigens from normal skin flora during this process.
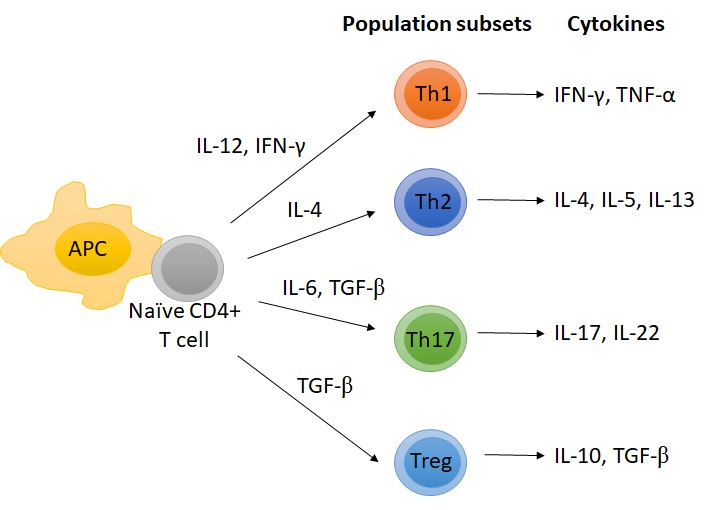
Figure 3: Polarization of naive T cells into different T helper cells.
The innate immune system and APCs play a role in triggering inflammation and initiating immune responses. LCs are sensitive to their microenvironment and can get rapidly activated by local inflammatory signals. Below are a few examples of the inflammatory skin disorders where LCs might play a causative role in either the initiation or the amplification of the inflammation.
Irritant Contact Dermatitis
Barrier disruption and irritant stress have been shown to activate LCs. Tape stripping experiments on human skin itself induce the release of inflammatory mediators from keratinocytes. This is exacerbated by the addition of irritants (e.g., sodium lauryl sulfate, acetone). The inflammatory microenvironment induces LCs activation and their migration outside the epidermis.28–30
Allergic Contact Dermatitis
Although it has been thought for a long time that LCs initiate allergic contact dermatitis and are responsible for activating T cells in the lymph nodes, recent studies investigating hapten-induced contact hypersensitivity in mice models depleting LCs, either constitutively or inducibly, could not identify essential roles for LCs in leading allergic responses.5 In these studies, dermal dendritic cells appeared as the main initiator of allergic reactions, while LCs could drive tolerogenic responses.30
Atopic Dermatitis (AD)
The origin of atopic dermatitis is still elusive, although its pathophysiology has been thoroughly investigated. Among causal factors, high allergic response and high circulating levels of immunoglobulin E (IgE) represent one hallmark of atopic dermatitis. LCs express Fc receptors at their surface. In atopic dermatitis, LCs express high levels of FcεRI, which recognize IgE-bound antigens. This activation results in subsequent inflammation leading to recruitment of members of the innate and adaptive immune systems (monocytes, eosinophils, T cells).31,32 A recent study, showed that LCs activated by a S. aureus strain isolated from atopic dermatitis patients favor the activation of Th2 lymphocytes.33 In addition, recent reports highlight a possible involvement of LCs in the activation of Th22 cells in acute and chronic atopic dermatitis lesions.25
Psoriasis
The role of epidermal LCs in the pathogenesis of psoriasis is unclear considering some of the contradictory studies published. Several mouse studies show that LCs or Langerin+ dendritic cells are not present in the inflammation of imiquimod-induced psoriasis,34 whereas other studies demonstrate that the absence of LCs leads to the aggravation of psoriasis features in double knock-out (DKO) mice.35 Conversely, in a imiquimod-induced psoriasis mouse model, LCs have been shown to produce IL-23, essential to activate a T cells subtype (γδ T cells) able to secrete IL-17, an important marker of psoriasis inflammation.36 Another study has shown that LCs activate IL-17-producing T cells through a presentation of self-lipid antigens on their CD1a receptor and contribute to psoriasis inflammation.37 Nevertheless, further investigations are required to clarify the role of LCs in psoriasis.
The skin is a dynamic organ and its immune function is gaining more and more importance as skin inflammatory diseases arise. One major goal of personal care product companies is to generate products able to re-equilibrate the skin and its barrier function, without inducing toxicity or an inflammatory response. To predict product sensitization, cell culture models have been established using immune cells. LCs represent good candidates as their activation is sensible to barrier disruption and local inflammation resulting from keratinocyte inflammation.
To model an in vitro assay of skin sensitization, a few models can be found in the literature and often use LC-like cells, which are easier to obtain than epidermal LCs. These LC-like cells are often obtained from primary blood precursors (CD34+ progenitors or CD14+ precursors) or from the MUTZ-3 cell line. After differentiation for approximately a week using appropriate cytokine cocktails (usually a combination of GM-CSF, IL-4, TGF-β, and/or TNF-α), these cells demonstrate LC features and can be treated with potent sensitizing products.
The sensitization potential is then determined by the ability to trigger LC-like cell activation by monitoring their migration and the upregulation of maturation markers. Most models are monolayer models, but a few skin models have been described.38,39 It is important to keep in mind that these cells, despite their resemblance to epidermal LCs, differ in their level of activation/reactivity compared to epidermal LCs.40 One should keep in mind that they represent a good model to predict a potential skin sensitization, but the use of these models might need to be reconsidered for other investigations.
LCs, which were originally mistaken with neuronal cells, are sentinels in the skin and are part of the first line of defense of the skin against pathogens, mediating signals of the innate and the adaptive immune system. In recent years, research on LC biology has improved knowledge on their origin, characteristics, and functions. Yet, LC physiology does not remain fully understood and it still needs to be clarified whether epidermal LCs contribute to local tissue activation of T cell subsets, why epidermal LCs are less reactive than their dermal counterparts, and what is the exact origin of epidermal LCs.
1. A. Kubo, K. Nagao, M. Yokouchi, H. Sasaki, and M. Amagai, External antigen uptake by Langerhans cells with reorganization of epidermal tight junction barrie, J. Exp. Med,. 206, 2937–2946 (2009).
2. M. Hitzler, A. Bergert, A. Luch, and M. Peiser, Evaluation of selected biomarkers for the detection of chemical sensitization in human skin: a comparative study applying THP-1, MUTZ-3 and primary dendritic cells in culture, Toxicol. In Vitro Int., 27, 1659–1669 (2013).
3. H. Tuschl, and R. Kovac, Langerhans cells and immature dendritic cells as model systems for screening of skin sensitizers, Toxicol. In Vitro, 15, 327–331 (2001).
4. S.W. Kashem, M. Haniffa, and D.H. Kaplan, Antigen-presenting cells in the skin, Annu. Rev. Immunol., 35, 469–499 (2017).
5. T. Doebel, B. Voisin, and K. Nagao, Langerhans cells – the macrophage in dendritic cell clothing, Trends Immunol., 38, 817–828 (2017).
6. D.A. Yanez, R.K. Lacher, A. Vidyarthi, and O.R. Colegio, The role of macrophages in skin homeostasis, Pflugers Arch., 469, 455–463 (2017).
7. D. Lipsker, U. Ziylan, R. McDermott, D. Spehner, F. Proamer, J.P. Cazenave, B. Goud, H. de la Salle, J. Salamero, and D. Hanau, Cored tubules are present in human epidermal Langerhans cells, J. Invest. Dermatol., 120, 407–410 (2003).
8. M.B.M. Teunissen, J. Wormmeester, S.R. Krieg, P.J. Peters, I.M.C. Vogels, M.L. Kapsenberg, and J.D. Bos, Human epidermal Langerhans cells undergo profound morphologic and phenotypical changes during in vitro culture, J. Invest. Dermatol., 94, 166–173 (1990).
9. J. Valladeau, C. Dezutter-Dambuyant, and S. Saeland, Langerin/CD207 sheds light on formation of bsirbeck granules and their possible function in Langerhans cells, Immunol. Res., 28, 93–107 (2003).
10. P. Stoitzner and N. Romani, Langerin, the 'Catcher in the Rye': an important receptor for pathogens on Langerhans cells, Eur. J. Immunol., 41, 2526–2529 (2011).
11. I. Van Rhijn, D.I. Godfrey, J. Rossjohn, and D.B. Moody, Lipid and small-molecule display by CD1 and MR1, Nat. Rev. Immunol., 15, 643–654 (2015).
12. H. Hirschberg, L.R. Braathen, and E. Thorsby, Antigen presentation by vascular endothelial cells and epidermal Langerhans cells: the role of HLA-DR, Immunol. Rev., 66, 57–77 (1982).
13. M.C. Udey, Cadherins and Langerhans cell immunobiology, Clin. Exp. Immunol., 107 Suppl 1, 6–8 (1997).
14. M.D. Fleming, J.L. Pinkus, M.V. Fournier, S.W. Alexander, C. Tam, M. Loda, S.E. Sallan, K.E. Nichols, D.F. Carpentieri, G.S. Pinkus, and B.J. Rollins, Coincident expression of the chemokine receptors CCR6 and CCR7 by pathologic Langerhans cells in Langerhans cell histiocytosis, Blood, 101, 2473–2475 (2003).
15. M. Merad, F. Ginhoux, and M. Collin, Origin, homeostasis and function of Langerhans cells and other langerin-expressing dendritic cells, Nat. Rev. Immunol., 8, 935–947 (2008).
16. N. Yasmin, T. Bauer, M. Modak, K. Wagner, C. Schuster, R. Köffel, M. Seyerl, J. Stöckl, A. Elbe-Bürger, D. Graf, and H. Strobl, Identification of bone morphogenetic protein 7 (BMP7) as an instructive factor for human epidermal Langerhans cell differentiation, J. Exp. Med., 210, 2597–2610 (2013).
17. Y. Wang, M. Bugatti, T.K. Ulland, W. Vermi, S. Gilfillan, and M. Colonna, Nonredundant roles of keratinocyte-derived IL-34 and neutrophil-derived CSF1 in Langerhans cell renewal in the steady state and during inflammation, Eur. J. Immunol., 46, 552–559 (2016).
18. V. Flacher, M. Bouschbacher, E. Verronèse, C. Massacrier, V. Sisirak, O. Berthier-Vergnes, B. de Saint-Vis, C. Caux, C. Dezutter-Dambuyant, S. Lebecque, and J. Valladeau, Human Langerhans cells express a specific TLR profile and differentially respond to viruses and Gram-positive bacteria, J. Immunol., 177, 7959–7967 (2006).
19. J.B. Frazão, P.R. Errante, and A. Condino-Neto, Toll-like receptors’ pathway disturbances are associated with increased susceptibility to infections in humans, Arch. Immunol. Ther. Exp., (Warsz.), 61, 427–443 (2013).
20. A.M. van der Aar, D.I. Picavet, F.J. Muller, L. de Boer, T.M. van Capel, S.A Zaat, J.D. Bos, H. Janssen, T.C George,M.L. Kapsenberg, S.M van Ham, M.B. Teunissen, and E.C de Jong, Langerhans cells favor skin flora tolerance through limited presentation of bacterial antigens and induction of regulatory T cells, J. Invest. Dermatol., 133, 1240–1249 (2013).
21. A.M. van der Aar, R. de Groot, M. Sanchez-Hernandez, E.W. Taanman, R.A. van Lier, M.B. Teunissen, E.C. de Jong, and M.L. Kapsenberg, Cutting edge: virus selectively primes human langerhans cells for CD70 expression promoting CD8+ T cell responses, J. Immunol., 187, 3488–3492 (2011).
22. C. Levin, H. Perrin, and B. Combadiere, Tailored immunity by skin antigen-presenting cells, Hum. Vaccines Immunother., 11, 27–36 (2015).
23. L. Furio, I. Briotet, A. Journeaux, H. Billard, and J. Péguet-Navarro, Human langerhans cells are more efficient than CD14(-)CD1c(+) dermal dendritic cells at priming naive CD4(+) T cells, J. Invest. Dermatol., 130, 1345–1354 (2010).
24. A.R. Mathers, B.M. Janelsins, J.P. Rubin, O.A. Tkacheva, W.J. Shufesky, S.C. Watkins, A.E. Morelli, and A.T. Larregina, Differential capability of human cutaneous dendritic cell subsets to initiate Th17 responses, J. Immunol., 182, 921–933 (2009).
25. H. Fujita, K.E. Nograles, T. Kikuchi, J. Gonzalez, J.A. Carucci, and J.G. Krueger, Human Langerhans cells induce distinct IL-22-producing CD4+ T cells lacking IL-17 production, Proc. Natl. Acad. Sci. U.S.A., 106, 21795–21800 (2009).
26. J. Seneschal, R.A. Clark, A. Gehad, C.M. Baecher-Allan, and T.S. Kupper,. Human epidermal Langerhans cells maintain immune homeostasis in skin by activating skin resident regulatory T cells, Immunity, 36, 873–884 (2012).
27. J. Banchereau, L. Thompson-Snipes, S. Zurawski, J.P. Blanck, Y. Cao, S. Clayton, J.P. Gorvel, G. Zurawski, and E. Klechevsky, The differential production of cytokines by human Langerhans cells and dermal CD14(+) DCs controls CTL priming. Blood, 119, 5742–5749 (2012).
28. K. Ouwehand, R.J. Scheper, T.D. de Gruijl, and S. Gibbs, Epidermis-to-dermis migration of immature Langerhans cells upon topical irritant exposure is dependent on CCL2 and CCL5, Eur. J. Immunol., 40, 2026–2034 (2010).
29. H. Tang, C. Schlapbach, A.S. Hassan, D. Simon, and N. Yawalkar, Characterization of dendritic cells and macrophages in irritant contact dermatitis, J. Dermatol. Sci., 57, 216–218 (2010).
30. J.K. Gittler, J.G. Krueger, and E. Guttman-Yassky, Atopic dermatitis results in intrinsic barrier and immune abnormalities: implications for contact dermatitis, J. Allergy Clin. Immunol., 131, 300–313 (2013).
31. N. Novak, T. Bieber, and D.Y.M. Leung, Immune mechanisms leading to atopic dermatitis, J. Allergy Clin. Immunol., 112, S128-S139 (2003).
32. G.C. Mudde, F.C. Van Reijsen, G.J. Boland, G.C. de Gast, P.L. Bruijnzeel, and C.A. Bruijnzeel-Koomen, Allergen presentation by epidermal Langerhans’ cells from patients with atopic dermatitis is mediated by IgE, Immunology, 69, 335–341 (1990).
33. K. Iwamoto, M. Moriwaki, Y. Niitsu, M. Saino, S. Takahagi, J. Hisatsune, M. Sugai, and M. Hide, Staphylococcus aureus from atopic dermatitis skin alters cytokine production triggered by monocyte-derived Langerhans cell, J. Dermatol. Sci., 88, 271–279 (2017).
34. C. Wohn, J.L. Ober-Blöbaum, S. Haak, S. Pantelyushin, C. Cheong, S.P. Zahner, S. Onderwater, M. Kant, H. Weighardt, B. Holzmann, B. Reizis, B. Becher, E.P. Prens, and B.E. Clausen, Langerin(neg) conventional dendritic cells produce IL-23 to drive psoriatic plaque formation in mice, Proc. Natl. Acad. Sci. U.S.A., 110, 10723–10728 (2013).
35. E. Glitzner, A. Korosec, P.M. Brunner, B. Drobits, N. Amberg, H.B. Schonthaler, T. Kopp, E.F. Wagner, G. Stingl, M. Holcmann, and M. Sibili, Specific roles for dendritic cell subsets during initiation and progression of psoriasis, EMBO Mol. Med., 6, 1312–1327 (2014).
36. R. Yoshiki, K. Kabashima, T. Honda, S. Nakamizo, Y. Sawada, K. Sugita, H. Yoshioka, S. Ohmori, B. Malissen, Y. Tokura, and M. Nakamura, IL-23 from Langerhans cells is required for the development of imiquimod-induced psoriasis-like dermatitis by induction of IL-17A-producing γδ T cells, J. Invest. Dermatol., 134, 1912–1921 (2014).
37. J.H. Kim, Y. Hu, T. Yongqing, J. Kim, V.A. Hughes, J. Le Nours, E.A. Marquez, A.W. Purcell, Q. Wan, M. Sugita, J. Rossjohn, and F. Winau, CD1a on Langerhans cells controls inflammatory skin disease, Nat. Immunol., 17, 1159–1166 (2016).
38. K. Ouwehand, S.W. Spiekstra, T. Waaijman, R.J. Scheper, T.D. de Gruijl, and S. Gibbs, Technical advance: Langerhans cells derived from a human cell line in a full-thickness skin equivalent undergo allergen-induced maturation and migration, J. Leukoc. Biol., 90, 1027–1033 (2011).
39. K. Ouwehand, D. Oosterhoff, M. Breetveld, R.J. Scheper, T.D. de Gruijl, and S. Gibbs, Irritant-induced migration of Langerhans cells coincides with an IL-10-dependent switch to a macrophage-like phenotype, J. Invest. Dermatol., 131, 418–425 (2011).
40. M. Peiser, R. Wanner, and G. Kolde, Human epidermal Langerhans cells differ from monocyte-derived Langerhans cells in CD80 expression and in secretion of IL-12 after CD40 cross-linking, J. Leukoc. Biol., 76, 616–622 (2004).