

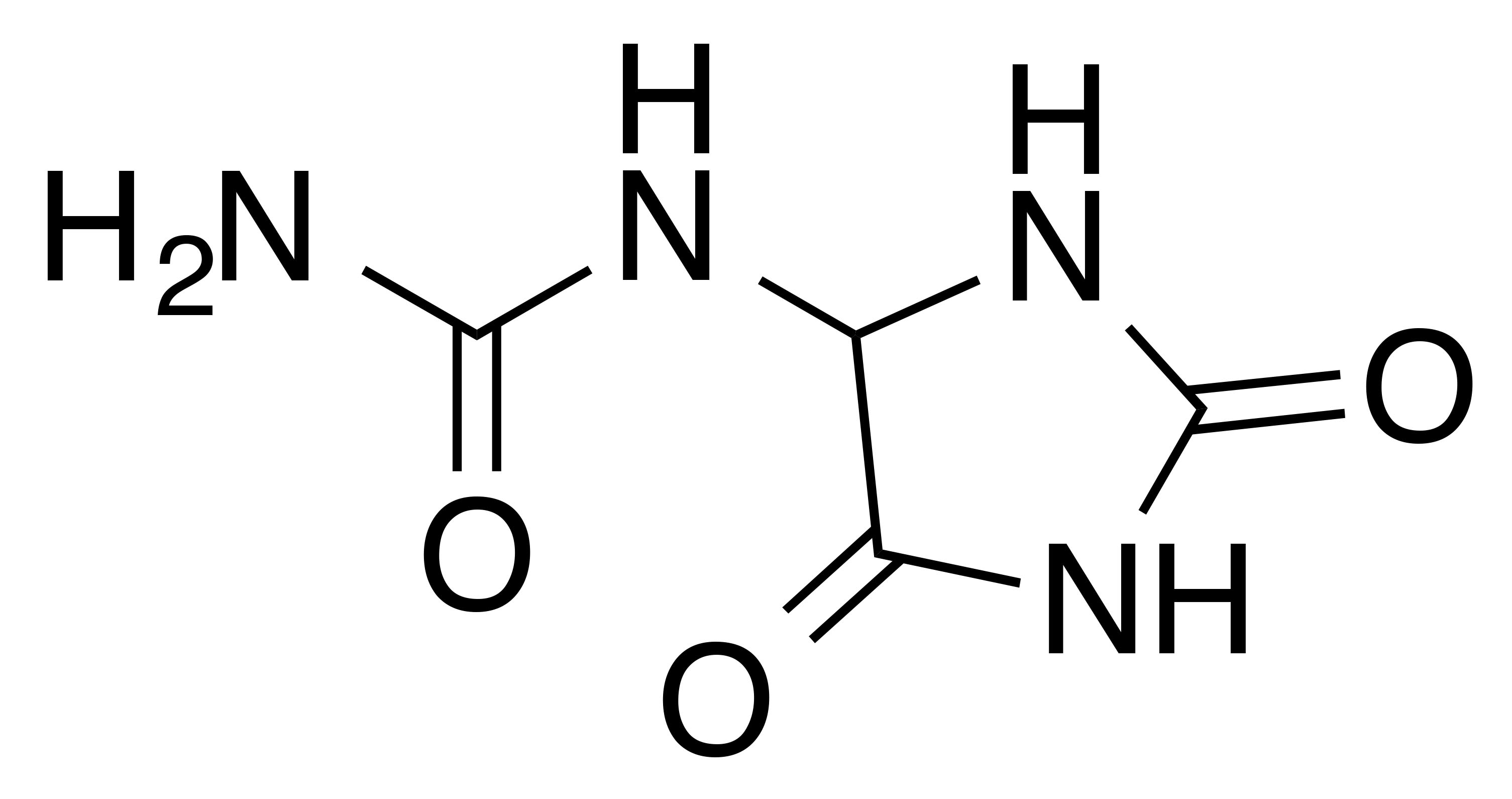
INCI name: allantoin
Synonyms: 5-ureidohydantoin, glyoxyldiureide, cordianine
Molecular formula: C4H6N4O3
Molecular weight: 158.116 g/mol
IUPAC name: (2,5-Dioxo-4-imidazolidinyl) urea
CAS number: 97-59-6
EC number: 202-592-8
In nature, allantoin is found in the comfrey plant (Symphytum officinale L.).1 It is also a metabolic product in many other plants, animals, and bacteria, forming as a result of the oxidation of uric acid. In humans and other primates, who lack the urate oxidase enzyme, the formation of uric acid is the final step in purine metabolism. While allantoin may be obtained from natural sources, it is normally prepared synthetically for cosmetic use, either by oxidation of uric acid, reaction of dichloroacetic acid and urea, or condensation reaction between glyoxylic acid and urea.2
Historically, allantoin was used to promote wound healing and treat skin ulcers.3 Nowadays, it is well recognized that allantoin has keratolytic properties in which it facilitates the desquamation of the stratum corneum. One of the key benefits of allantoin, in respect to other desquamation agents, is that it is non-irritating to the skin.3 Allantoin is typically used at concentrations of 0.0001% – 2.0% (w/w) in cosmetic formulations. In fact, allantoin is an FDA approved skin protectant that is approved for use in over-the-counter medications at 0.5% – 2.0%.4
Properties
Melting point: 238 °C5
Solubility: water (1 g/190 mL) and ethanol (1 g/500 mL); solubility increases with elevated temperature.5
References
1. R.M. Di Salvo, “Allantoin” in The Chemistry and Manufacture of Cosmetics: Vol. III – Ingredients, Book One, 3rd ed., Ed. M.L. Schlossman, 2002.
2. Allantoin, A Safe and Effective Skin Protectant, Akema Fine Chemicals, product literature, accessed June 2017, www.akema.it.
3. Allantoin, An Impressive History, DSM Nutritional Products, product literature, accessed June 2017, www.dsmnutritionalproducts.com.
4. L.C. Becker, W.F. Bergfeld, D.V. Belsito, C.D. Klaassen, J.G. Marks, R.C. Shank, T.J. Slaga, P.W. Snyder, and F. Alan Andersen, Final report of the safety assessment of allantoin and its related complexes, Int. J. Toxicol., 29(3 Suppl), 84S-97S (2010).
5. M.J. O’Neil, The Merck Index, 15th ed., The Royal Society of Chemistry: Cambridge, UK (2013).
Further Reading
1. X.B. Chen, W. Matuszewski, and J. Kowalczyk, Determination of allantoin in biological, cosmetic, and pharmaceutical samples, J. AOAC Int., 79, 628-635 (1996).
2. Z.R. Zaidi, F.J. Sena, and C.P. Basilio, Stability assay of allantoin in lotions and creams by high-pressure liquid chromatography, J. Pharm. Sci., 71, 997-999 (1982).