

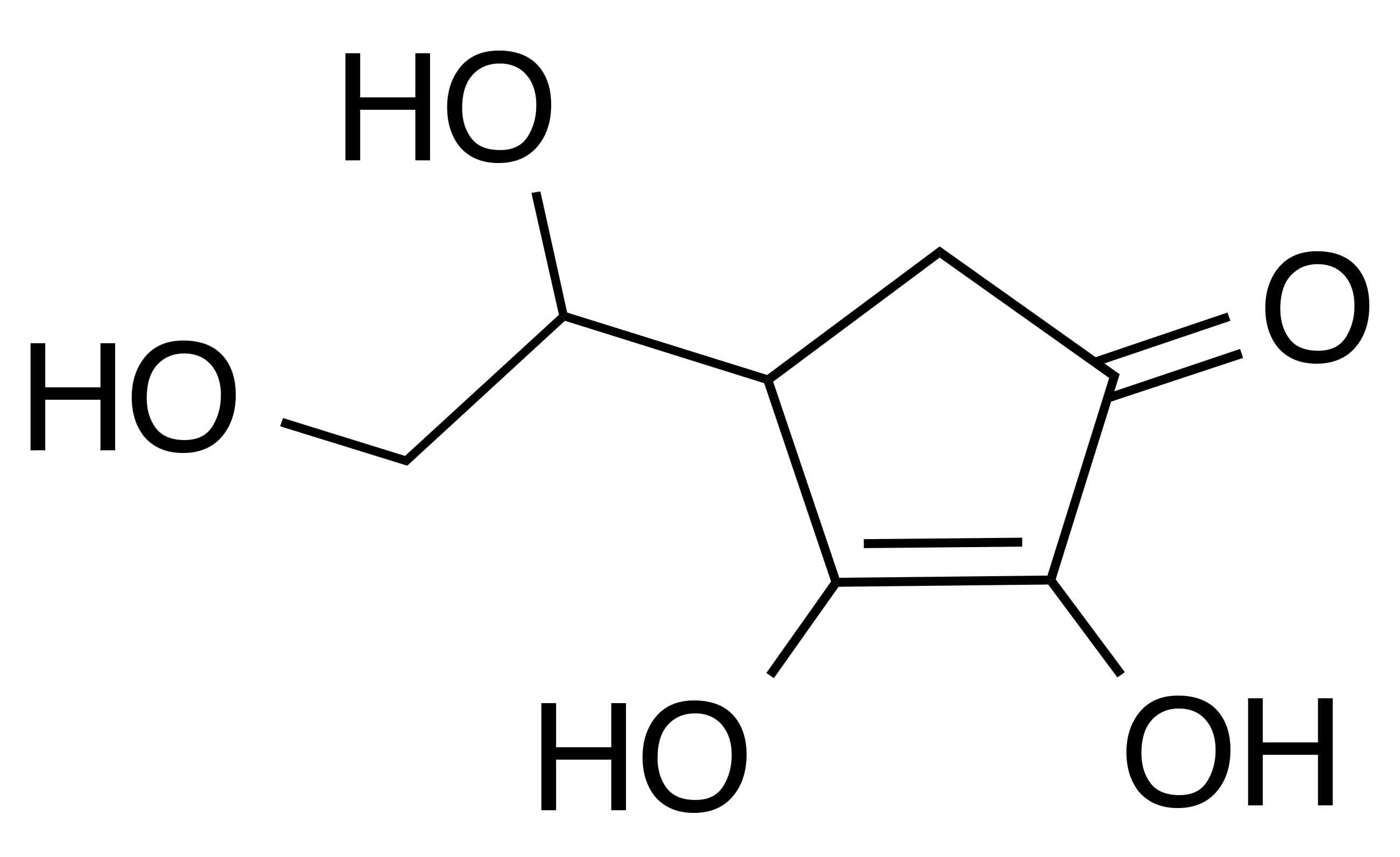
Synonyms: vitamin C; L-ascorbic acid
Molecular formula: C6H8O6
Molecular weight: 176.12 g/mol
IUPAC name: (2R)-2-[(1S)-1,2-dihydroxyethyl]-3,4-dihydroxy-2H-furan-5-one
CAS number: 50-81-7
EC number: 200-066-2
Ascorbic acid is an antioxidant that is found naturally in many fruits and vegetables. Some animals (e.g., the goat, cat, dog, etc.) are able to endogenously synthesize ascorbic acid. Humans and other animals, through an evolutionary genetic defect, do not express the enzyme (gulonolactone oxidase) that catalyzes the last step in the biosynthesis of vitamin C.1 Therefore, these animal species must obtain ascorbic acid (or vitamin C) through the diet. Apart from its antioxidant properties, vitamin C is also essential for proper biosynthesis of collagen. Interestingly, lack of dietary intake vitamin C can lead to scurvy—a disease that was common among European explorers in the 17th and 18th centuries during long periods at sea without access to fresh fruits and vegetables. It should be noted that scurvy is still around today, although not nearly as prevalent as it used to be, with cases reported mostly in developing countries.
Numerous studies have demonstrated the positive health benefits of ascorbic acid in the topical treatment of skin.2,3 Mostly, this is in the context of the prevention of erythema, photoaging, and photoimmunosuppression.2 In addition, ascorbic acid has anti-tyrosinase activity, which has lead to its use in skin whitening preparations.4,5 While there are many positive benefits for the use of ascorbic acid as a topical agent for skin care, its inclusion in cosmetic formulations is often hampered by stability issues. To circumvent such issues, many ascorbic acid derivatives with improved stability are available to cosmetic formulators, or ascorbic acid is formulated with other ingredients to help stabilize it.6 It should be noted, however, the efficacy of modified forms of ascorbic acid tend to not have equivalent antioxidant power as the parent compound. Another strategy is to encapsulate ascorbic acid in delivery vesicles that allow it to penetrate the skin, while maintaining it in its stable form.7
Properties8
Physical: white crystals or powder
Melting point: 190-192 °C
pKa: pK1= 4.17; pK2=11.57
Solubility: 1 g/3 mL H2O; 1 g/30 mL ethanol; 1 g/100 mL glycerol
λmax: 245 nm (pH 2); 265 nm (pH 6.4)
Stability: stable in air; oxidation occurs rapidly in H2O
References
1. G. Drouin, J.-R. Godin, and B. Pagé, The genetics of vitamin C loss in vertebrates, Curr. Genomics, 12, 371-378 (2011).
2. R.L. McMullen, Antioxidants and the Skin, Allured Books: Carol Stream, IL (2013).
3. P.K. Farris, Topical vitamin C: a useful agent for treating photoaging and other dermatologic conditions, Dermatol. Surg., 31(7 Pt 2), 814-817 (2005).
4. P.S. Telang, Vitamin C in dermatology, Indian Dermatol. Online J., 4, 143-146 (2013).
5. F. Al-Niaimi and N.Y.Z. Chiang, Topical vitamin C and the skin: mechanisms of action and clinical applications, J. Clin. Aesthet. Dermatol., 10, 14-17 (2017).
6. M.A. Sheraz, S. Ahmed, I. Ahmad, R.H. Shaikh, F.H.M. Vaid, and K. Iqbal, Formulation and stability of ascorbic acid in topical preparations, Syst. Rev. Pharm., 2, 86-90 (2011).
7. G. Serrano, P. Almudever, J.M. Serrano, J. Milara, A. Torrens, I. Exposito, and J. Cortijo, Phosphatidylcholine liposomes as carriers to improve topical ascorbic acid treatment of skin disorders, Clin. Cosmet. Investig. Dermatol., 8, 591-599 (2015).
8. M.J. O’Neil, The Merck Index, 15th ed., The Royal Society of Chemistry: Cambridge, UK (2013).