

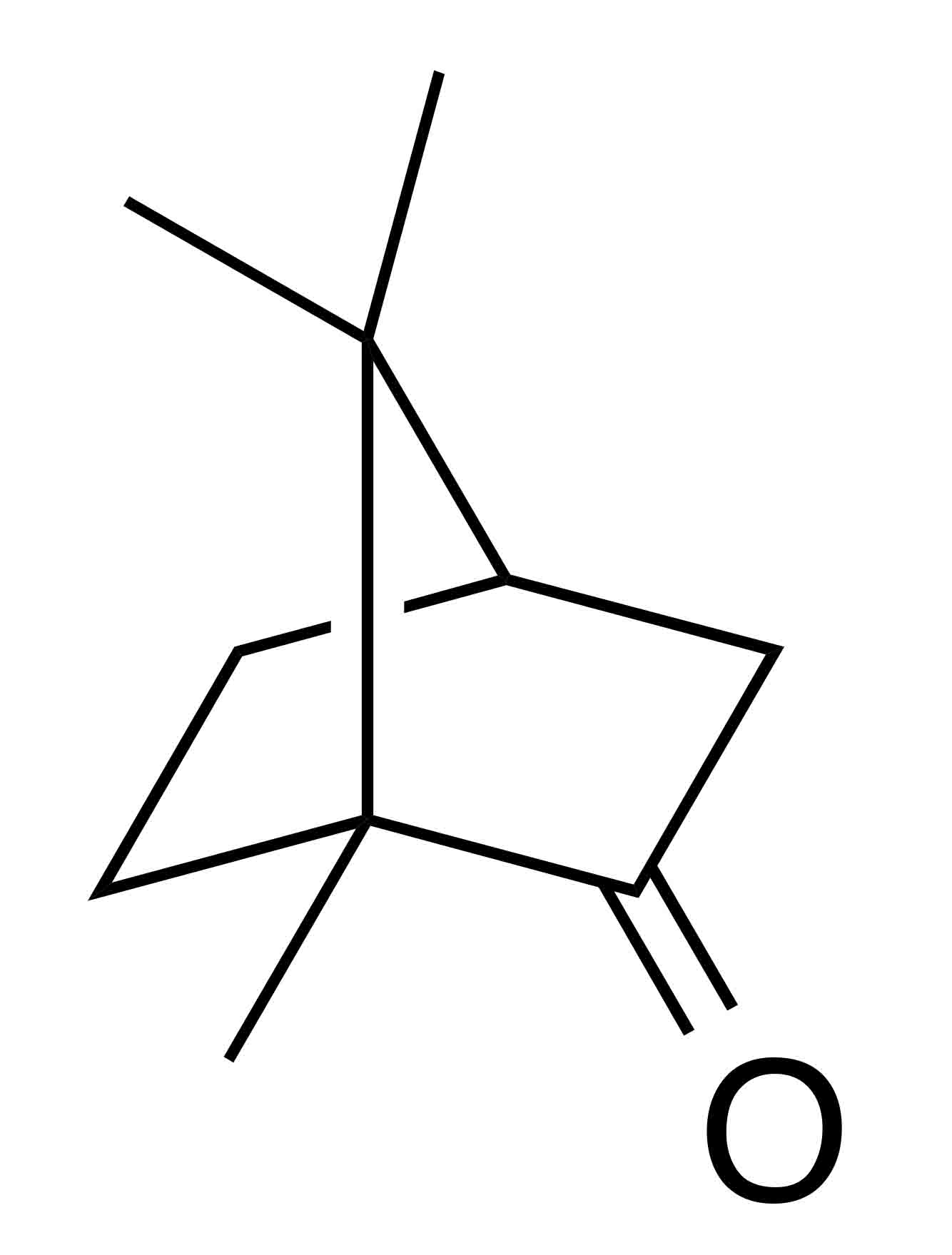
INCI name: Camphor oil, cinnamomum camphora (camphor) bark oil
Synonyms: DL-camphor, gum camphor, 2-bornanone
Molecular formula: C10H16O
Molecular weight: 152.24 g/mol
IUPAC name: 4,7,7-trimethylbicyclo[2.2.1]heptan-3-one
CAS number: 8008-51-3
EC number: 244-350-4
Camphor is a terpenoid isolated from the wood of the camphor laurel tree (Cinnamomum camphora). The natural form camphor is D-camphor, while the synthetic version is a mixture of the D and L enantiomers. Camphor has a long history of use as an antiseptic, analgesic, antipruritic, and counterirritant agent.1 It is also used as a decongestant and cough suppressant in the form of ointments, oils, and inhalants.2
When applied to skin, camphor initially causes a cooling sensation, which is followed by a localized heating sensation, resulting in an increase in blood flow to the site of treatment. Recent research has focused on understanding the mechanism of the activation of heat-sensitive receptors, specifically two members of transient receptor potential (TRP) channel superfamily: heat-sensitive TRP vanilloid subtype 1 (TRPV1) and 3 (TRPV3).3 It should be noted that camphor is often compared to menthol; however, menthol only provides a cooling sensation (there is no warming sensation).
To better understand the properties of camphor, researchers at Medical University of Gdansk in Poland used a tape-stripping technique coupled with gas chromatography to monitor penetration of camphor into the skin.4 Their findings suggest that the penetration of camphor into the skin depends on the delivery vehicle. For example, in a hydrogel formulation, penetration was found to be greater than in an oil solution.
In cosmetics, camphor oil is employed as a principal ingredient of bath products (e.g., a bath soak), massage oils, toners, facial and body cleaners, aromatherapy products, and perfumes.
Properties
Melting point: 174-179 °C5
Boiling point: 209 °C6
Solubility: At 25 °C, one gram dissolves in about 800 mL water, in 1 mL ethanol, 1 mL ether, and 0.5 mL chloroform. It is also soluble in concentrated mineral acids, phenol, liquid ammonia, and liquid sulfoxide.6
Density: 0.992 at 25 °C/4 °C6
Max UV/Vis absorption: 292 nm (chloroform)6
Refractive index: 1.54627
References
1. P. Zuccarini and G. Soldani, Camphor: benefits and risks of a widely used natural product, Acta Biologica Szegediensis, 53,77-82 (2009).
2. T. Kotaka, S. Kimura, M. Kashiwayanagi, and J. Iwamoto, Camphor induces cold and warm sensations with increases in skin and muscle blood flow in human, Biol. Pharm. Bull., 37, 1913–1918 (2014).
3. H. Xu, N.T. Blair, and D.E. Clapham, Camphor activates and strongly desensitizes the transient receptor potential vanilloid subtype 1 channel in a vanilloid-independent mechanism, J. Neurosci., 25, 8924-8937 (2005).
4. K. Cal, Skin disposition of d-camphor and l-menthol alone and together, Methods Find. Exp. Clin. Pharmacol., 31, 237-240 (2009).
5. R.J. Lewis, Hawley's Condensed Chemical Dictionary, 15th ed, John Wiley & Sons, Inc.: New York, NY, 2007.
6. M.J. O’Neil, The Merck Index, 15th ed., The Royal Society of Chemistry: Cambridge, UK (2013).
7. W.M. Haynes, Handbook of Chemistry and Physics, 97th ed., CRC Press: Boca Raton, FL (2016).