

INCI name: Ricinus communis (castor) seed oil
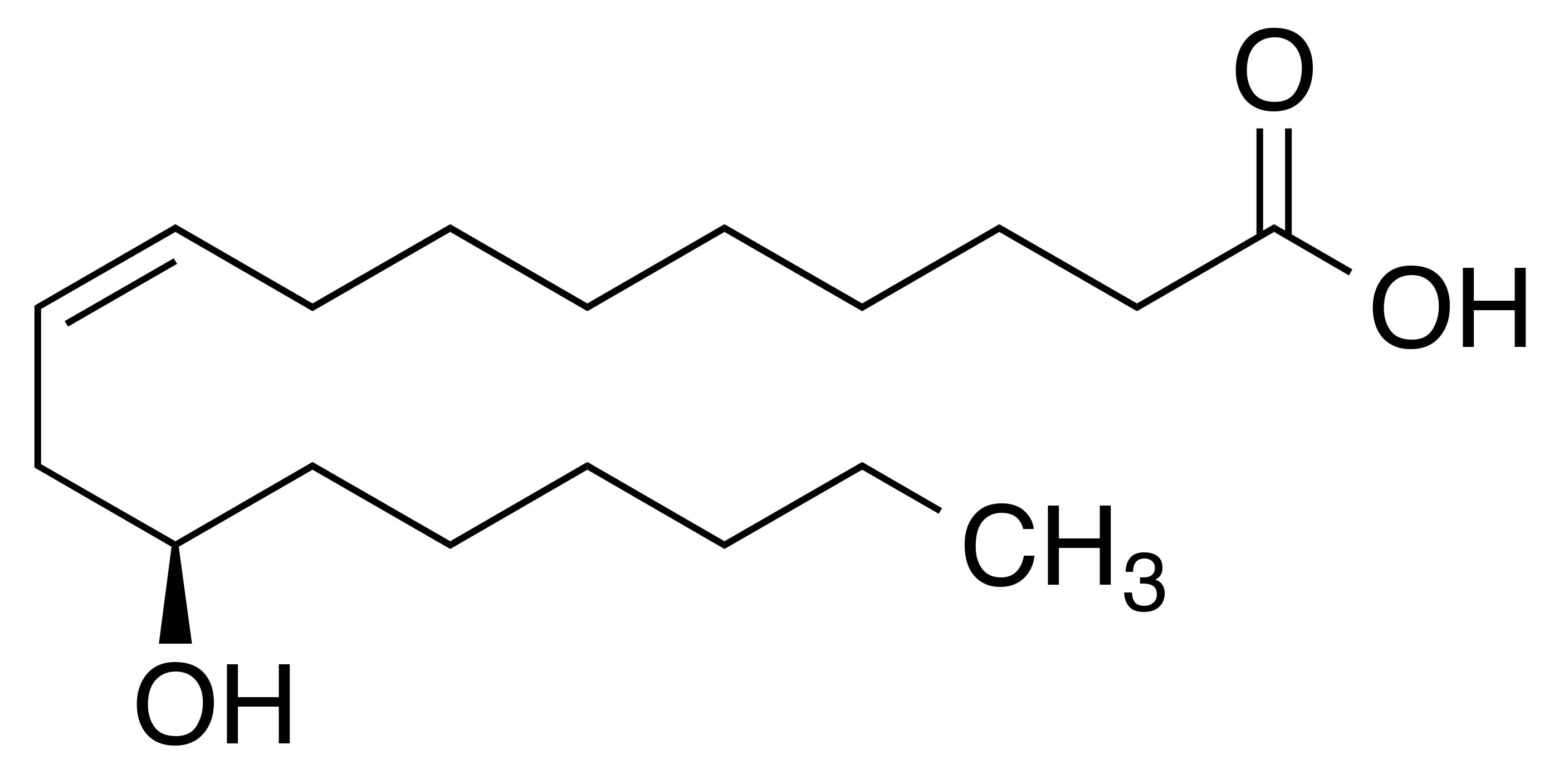
Synonyms: (R)-12-Hydroxy-cis-9-octadecenoic acid; 12-hydroxyoleic acid; ricinelaidic acid
Molecular formula: C18H34O3
Molecular weight: 298.47 g/mol
IUPAC name: (9Z,12R)-12-hydroxyoctadec-9-enoic acid
CAS number: 141-22-0
EC number: 205-470-2
Ricinoleic acid is the primary constituent of castor oil, which comes from the seeds of the castor bean plant (Ricinus communis L.).1 Approximately 85-90% of castor oil consists of ricinoleic acid. It should be noted that with such a high percentage of ricinoleic acid in castor oil, essentially it is a technical grade product—something that rarely happens in nature with plant oils. India is by far the largest producer of castor oil followed by China, Brazil, Thailand, Ethiopia, and Paraguay.2
Ricinoleic acid has a unique molecular structure attributed to the hydroxyl group located in the middle of the unsaturated chain. In the triglycerol form, hydrogen bonding occurs with the hydroxyl groups resulting in greater viscosity than in other oils. For this reason, castor oil is a superior lubricant as compared to other vegetable oils.1 In addition, the hydroxyl group—only separated from the double bond by a methylene group—imparts increased oxidative stability, thereby providing castor oil a longer shelf-life.3 Furthermore, this structural feature also affects the solvation properties and miscibility of ricinoleic acid. Unlike most fatty acids, ricinoleic acid is soluble in more polar solvents (e.g., alcohols).
Castor oil is used in many diverse areas including cosmetics, coatings, detergents, food, medicine, and plasticizer technology. Its traditional use as a household remedy has been known for many years to treat constipation, condition hair and skin, and alleve arthritis and muscle pain. During the middle and early twentieth century In the United States, many parents used castor oil to deter their children from misbehaving. It was common to threaten, “If you don’t behave, you are going to have to take your castor oil.” This was even famously portrayed in the cartoon series Tom and Jerry in a 1953 episode.4
In cosmetics, castor oil is found in variety of products including hair preparations, soaps, and skin ointments. In addition, one of the largest applications of castor oil in personal care is in lipstick preparations. In fact it has been quoted: “Castor oil is one of the stalwarts of lipstick formulation since it creates the highest gloss of all natural oils when applied to the skin. It is occlusive, water-repellant, tacky, and very protective to the skin.”5 It is found in high concentrations in lipsticks. There are also a number of ricinoleic acid derivatives used in a variety of personal care formulations including cetyl ricinoleate, ethyl ricinoleate, glyceryl ricinoleate, and zinc ricinoleate as well as polyethoxylated esters of ricinoleic acid. Overall, these materials are deemed safe for use in cosmetics.6 More than likely, the broad use of castor oil in cosmetics comes from the compatibility of ricinoleic acid with polar and nonpolar ingredients.
Properties
Viscosity: 889 cS3
Density: 0.959 g/mL3
Thermal conductivity: 4.727 W/m°C3
Boiling point: 245 °C7
Melting point: -2 to -5 °C3
LogP: log Kow = 6.19 (est)8
Refractive index: 1.4803
Solubility: alcohol, acetone, ether, chloroform7
Footnotes
1. T.A. McKeon, “Castor (Ricinus communis L.)” in Industrial Oil Crops, Eds. T.A. McKeon, D.G. Hayes, D.F. Hildebrand, and R.J. Weselake, AOCS Press: Champaign-Urbana, IL, USA, 75-112 (2016).
2. World Agroforestry Research Report (2009), Jatropha Reality Check: A field assessment of the agronomic and economic viability of Jatropha and other oil seed crops in Kenya. Published by Deutsche Gesellschaft fur Technische Zusammernarbeit (GTZ) Eschborn Germany.
3. V.R. Patel, G.G. Dumancas, L.C.K. Viswanath, R. Maples, and B..J. Subong, Castor oil: properties, uses, and optimization of processing parameters in commercial production, Lipid Insights, 9, 1-12 (2016). Please note that the physical properties provided are for castor oil, not pure ricinoleic acid.
4. Tom and Jerry, Baby Puss, Episode 12, Directed by William Hanna and Joseph Barbera and produced by Fred Quimby. It was released to theaters on December 25, 1943 by Metro-Goldwyn Mayer. https://www.youtube.com/watch?v=-84n9X8nKMI.
5. A. Dweck, “Use of Botanicals in Cosmetics” in Harry’s Cosmeticology, 8th ed., Ed. M.M. Rieger, Chemical Publishing: New York, NY, USA (2000).
6. CIR Expert Panel, Final report on the safety assessment of Ricinus Communis (Castor) Seed Oil, Hydrogenated Castor Oil, Glyceryl Ricinoleate, Glyceryl Ricinoleate SE, Ricinoleic Acid, Potassium Ricinoleate, Sodium Ricinoleate, Zinc Ricinoleate, Cetyl Ricinoleate, Ethyl Ricinoleate, Glycol Ricinoleate, Isopropyl Ricinoleate, Methyl Ricinoleate, and Octyldodecyl Ricinoleate, Int. J. Toxicol., 26 (Suppl. 3), 31-77 (2007).
7. M.J. O’Neil, The Merck Index, 15th ed., The Royal Society of Chemistry: Cambridge, UK (2013).
8. US EPA; Estimation Program Interface (EPI) Suite. Ver. 4.0. Jan, 2009. Available from, as of May 21, 2010: http://www.epa.gov/oppt/exposure/pubs/episuitedl.htm.