

INCI name: parfum
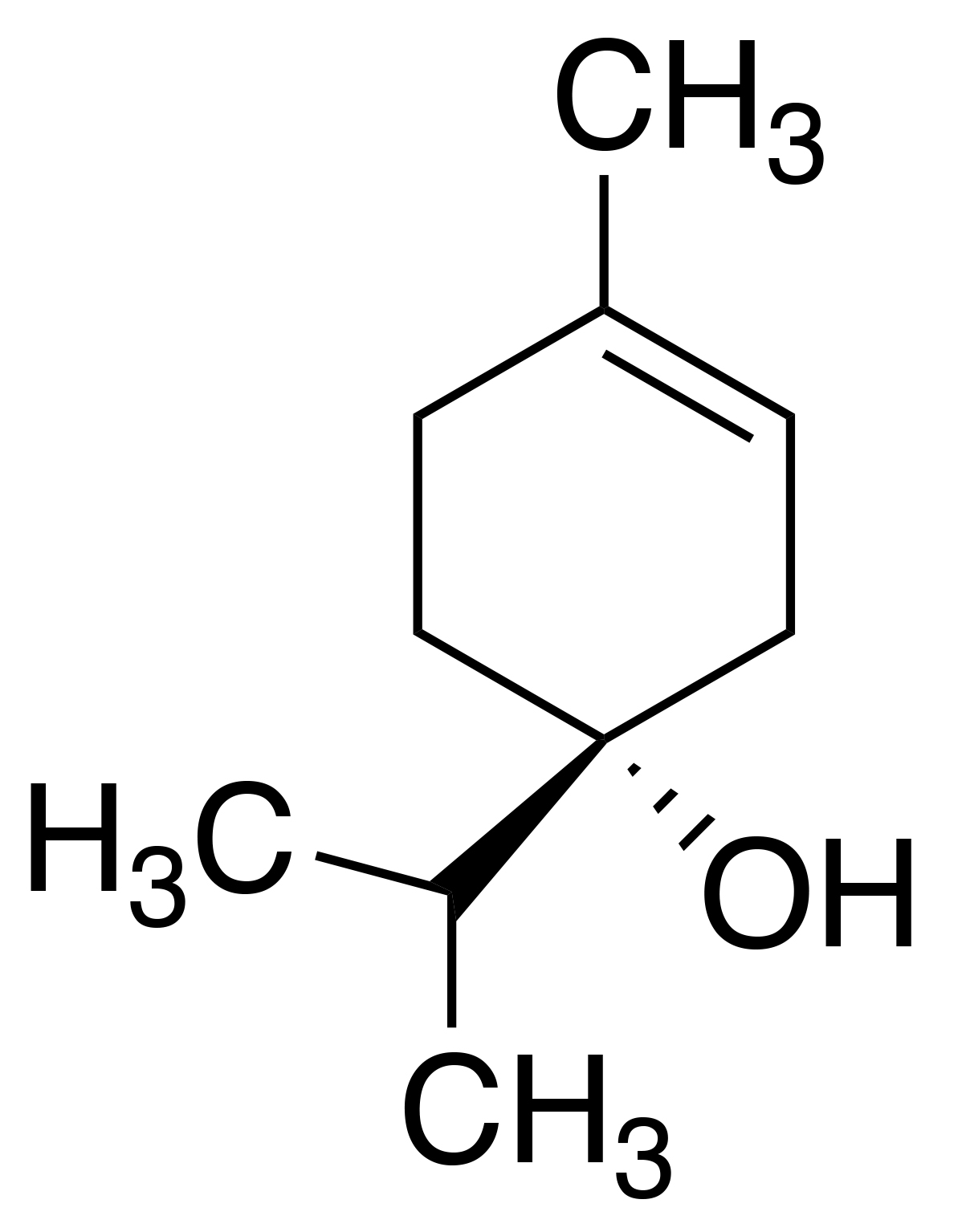
Synonyms: 3-cyclohexen-1-ol; 4-terpineol
Molecular formula: C10H18O
Molecular weight: 154.25 g/mol
IUPAC name: 4-methyl-1-(propan-2-yl)cyclohex-3-en-1-ol
CAS number: 562-74-3
EC number: 629-645-1
Terpinen-4-ol (a monocyclic terpene alcohol) is the primary component of the essential oil of Melaleuca alternifolia (tea tree oil), typically representing about 40% of the total oil composition.1 The tea tree is native to Australia and the essential oil is obtained from its leaves. It is commonly used as a traditional herbal medicine to treat the skin for acne and dandruff as well as bacterial and fungal infections.2,3 Terpinen-4-ol provides the broad antimicrobial activity and anti-inflammatory properties of tea tree oil. In cosmetics, tea tree oil has become a popular ingredient in shampoos, skin and nail creams, and massage oils.4 The odor of tea tree oil is similar to camphoraceous essential oils such as camphor, eucalyptus, frankincense, rosemary, and sage. When applied to skin it provides a cooling sensation.
Properties
Boiling point: 209 °C5
Melting point: 137-188 °C5
Density: 0.9275 g/cm3 (15 °C)5
Solubility: 1,491 ppm6
Log KOW: 3.267
References
1. C. Carson, K. Hammer, and T. Riley, Melaleuca alternifolia (tea tree) oil: a review of antimicrobial and other medicinal properties, Clin. Microbiol. Rev., 19(1), 50-62 (2006); https://doi.org/10.1128/CMR.19.1.50-62.2006.
2. N. Pazyar, R. Yaghoobi, N. Bagherani, and A. Kazerouni, A review of applications of tea tree oil in dermatology, Int. J. Dermatol., 52(7), 784-790 (2013); https://doi.org/10.1111/j.1365-4632.2012.05654.x.
3. J. Reichling, U. Landvatter, H. Wagner, K. Kostka, and U. Schaefer, In vitro studies on release and human skin permeation of Australian tea tree oil (TTO) from topical formulations, Eur. J. Pharm. Biopharm., 64(2), 222-228 (2006); https://doi.org/10.1016/j.ejpb.2006.05.006.
4. D. Larson and S. Jacob, Tea tree oil, Dermatitis, 23(1), 48-49 (2012); doi: 10.1097/DER.0b013e31823e202d.
5. Terpinen-4-ol. CAS Common Chemistry. CAS, a division of the American Chemical Society; https://commonchemistry.cas.org/detail?cas_rn=562-74-3; (retrieved 2022-10-05) (CAS RN: 562-74-3).
6. S. Griffin, S. Wyllie, J. Markham, and D. Leach, The role of structure and molecular properties of terpenoids in determining their antimicrobial activity, Flav. Fragr. J., 14, 322-332 (1999); https://doi.org/10.1002/(SICI)1099-1026(199909/10)14:5<322::AID-FFJ837>3.0.CO;2-4.
7. S. Griffin, S. Wyllie, and J. Markham, Determination of octanol-water partition coefficients for terpenoids using reversed-phase high-performance liquid chromatography, J. Chromatogr. A, 864, 221-228 (1999); https://doi.org/10.1016/S0021-9673(99)01009-2.