

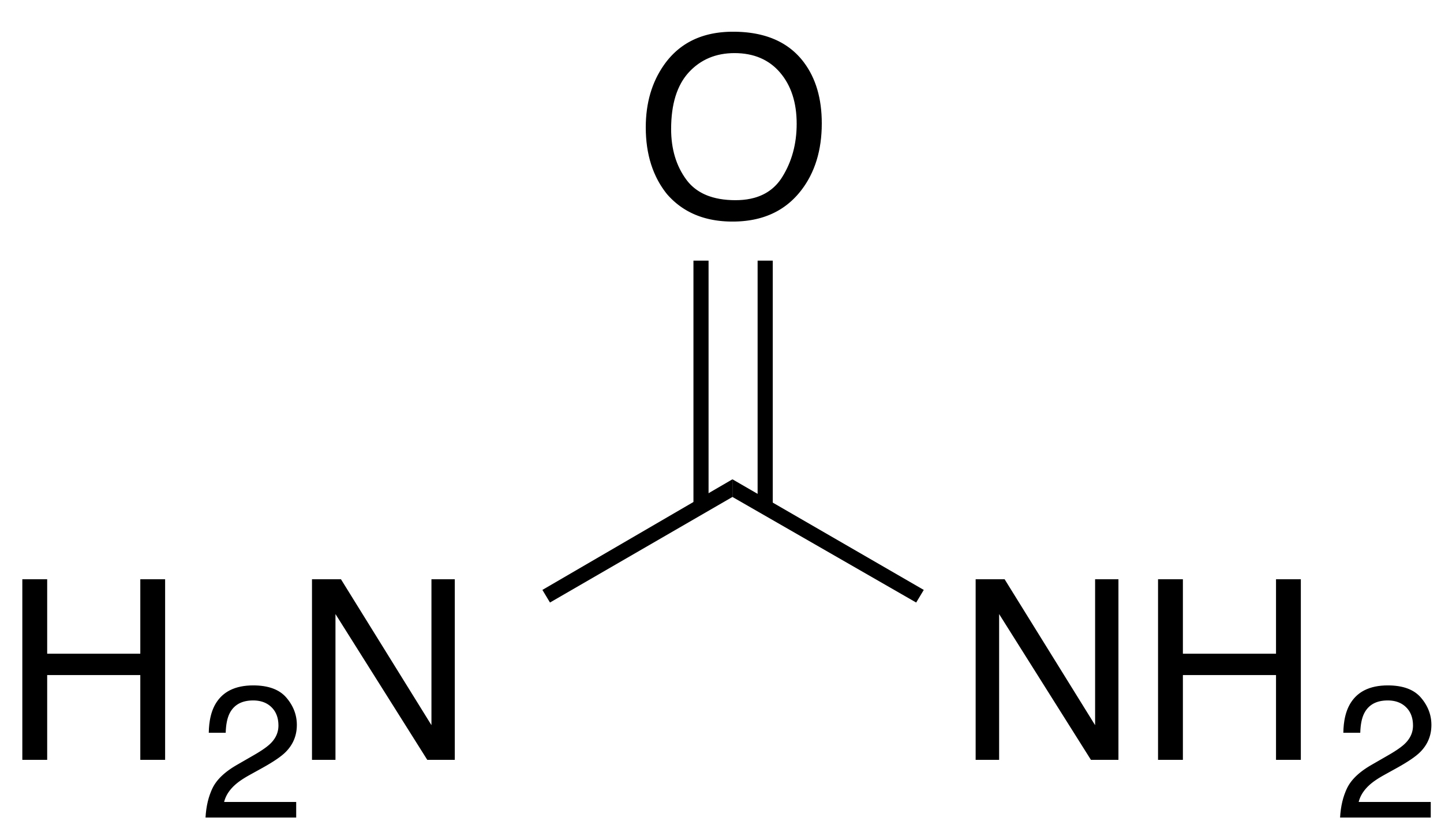
INCI name: urea
Synonyms: carbamide, carbonyldiamide, isourea
Molecular formula: CH4N2O
Molecular weight: 60.06 g/mol
IUPAC name: diaminomethanal
CAS number: 57-13-6
EC number: 200-315-5
Urea is a product of protein metabolism and is characterized by two primary amine groups that are attached by a carbonyl group. It is the principal solid component of urine and is also found in other bodily fluids. In skin, urea is a degradation product of proteins and serves as one of the components of the natural moisturizing factor. It is a very hygroscopic compound that attracts water and maintains a moisturized stratum corneum. Interestingly, patients with atopic dermatitis and other dry skin conditions have reduced levels of urea in their stratum corneum.1 In addition, urea is secreted by eccrine sweat glands, forming part of the acid mantle at the outermost surface of the skin.
Urea has a long history in traditional medicine and was often used to treat infections.2 Nowadays, urea is prepared synthetically and is found in a number of topical formulations. It is an important component of many types of skin care products, especially moisturizers, due to its humectant and barrier-improving properties. In addition, it is efficacious in treating a number of skin ailments including xerosis, ichthyosis, psoriasis, and several keratoses.3 In over-the-counter products urea concentrations are generally around 5-10%. In some cases, urea is found in prescription formulations at concentrations as high as 50%; however, such treatments are normally intended for treating nail disorders.4
Properties
Density: 1.32 g/mL5
Melting point: 132.7 °C6
Refractive index: 1.4845
Solubility: 1 g of urea dissolves in 1 mL water, 10 mL 95% alcohol, 20 mL absolute alcohol, and 2 mL glyercol6
References
1. K. Wellner and W. Wohlrab, Quantitative evaluation of urea in stratum corneum of human skin, Arch. Dermatol. Res., 285, 239-240 (1993).
2. M. Lodén, The use of urea in the treatment of dry skin, In Treatment of Dry Skin Syndrome: The Art and Science of Moisturizers, Springer-Verlag: Berlin, pp. 481-492, 2012.
3. N.S. Scheinfeld, Urea: a review of scientific and clinical data, Skinmed, 8(2), 102-106 (2010).
4. M. Pan, G. Heinecke, S. Bernardo, C. Tsui, and J. Levitt, Urea: a comprehensive review of the literature, Dermatol. Online J., 19(11), http://escholarship.org/uc/item/11x463rp (2013).
5. W.M. Haynes, Handbook of Chemistry and Physics, 97th ed., CRC Press: Boca Raton, FL (2016).
6. M.J. O’Neil, The Merck Index, 15th ed., The Royal Society of Chemistry: Cambridge, UK (2013).