

By The Cosmetic Chemist Staff
December 15, 2016

Fullerenes represent one type of allotrope of carbon.1 The most common form is a C60 molecule containing a hollow molecular sphere that is characterized by its highly conjugated structure consisting of 30 carbon double bonds (Figure 1). Fullerenes were discovered in 1985 by researchers at Rice University in Houston, TX (United States), who later won the Nobel Prize in Chemistry.2,3 The fullerenes became known as Buckminster Fullerenes, or Buckyballs, and received their name due to their structural resemblance to a geodesic sphere, specifically the Montréal Biosphère, which was designed by the American Architect, Buckminster Fuller.
Fullerenes are prepared by vaporizing carbon, which takes place by generating a current between two carbon rods, and then allowing it to condense in an inert gas. They have unique physicochemical properties and offer much promise in biomedical applications. The conjugated nature of the fullerene sphere provides it with free radical scavenging properties allowing it to act like a free radical sponge.4,5 Its antioxidant properties have been exploited in several studies focused on understanding how fullerenes can combat free radical damage in keratinocyte cell cultures exposed to UV radiation.6-8
Since C60 is very hydrophobic, a focus of contemporary fullerene research is to increase the degree of hydrophilicity of the molecule to make it more biologically available. One approach is to functionalize the outersurface of the fullerene sphere with hydroxyl groups—yielding compounds known as fullerenols (polyhydroxylated fullerenes). Additional chemical modifications that increase the hydrophilicity of fullerenes include the introduction of poly(ethylene glycol) units to the outer surface of the molecule, or the incorporation of the fullerene into a poly(vinylpyrrolidone) or γ-cyclodextrin shell. Finally, encapsulation of fullerenes into liposomes is yet another approach to increasing the solubility of fullerenes in water. All of these avenues have been successful at increasing the efficacy of fullerene in preventing free radical damage in keratinocyte cell culture systems.6-8
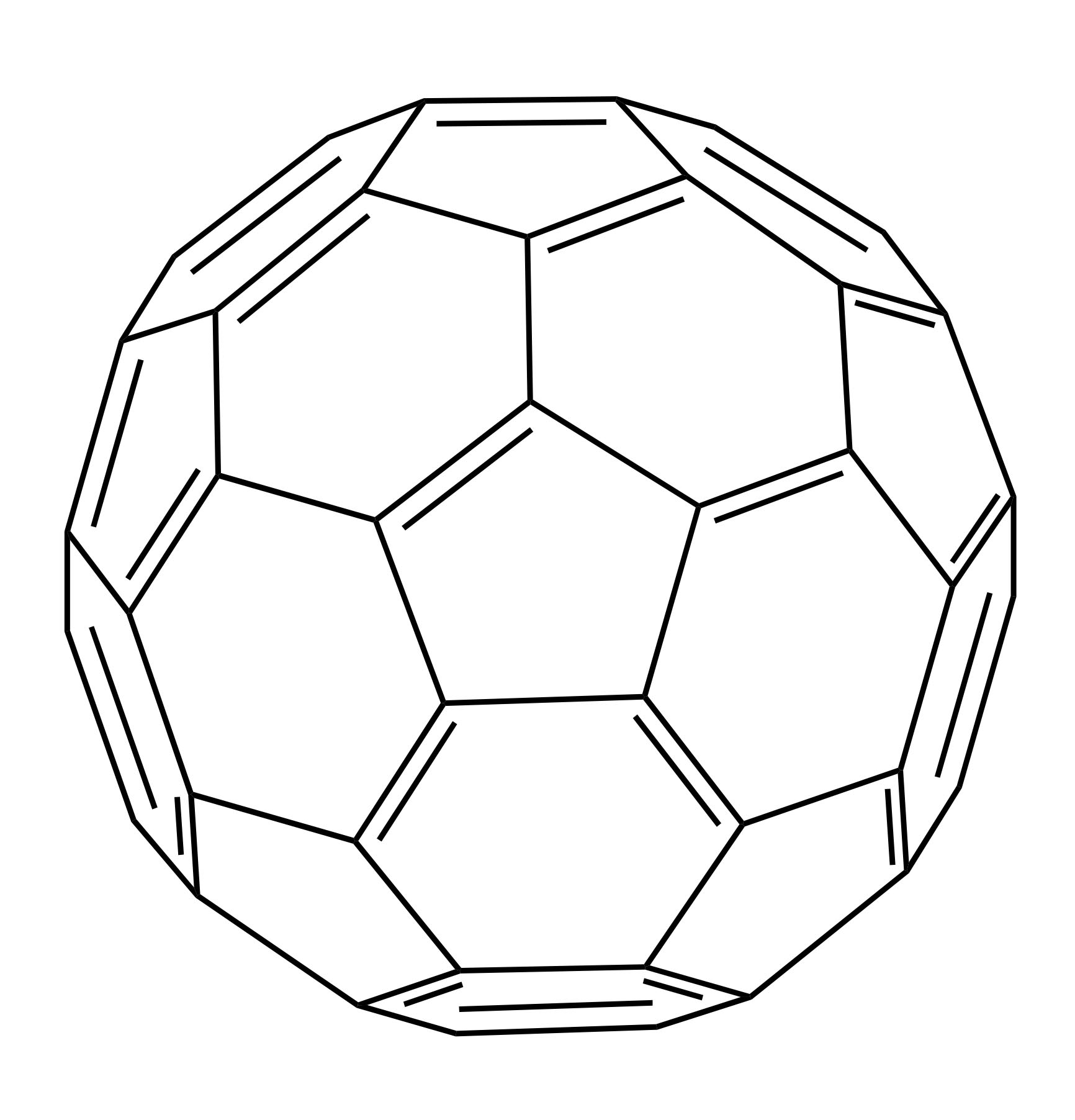
Figure 1: Molecular structure of C60 fullerene.
Several other studies have actually examined the penetration of C60 fullerene derivatives into the skin. For example, utilizing confocal Raman microscopy, researchers from North Carolina State University and Rice University observed the penetration into ex vivo skin of C60 fullerene modified with small peptide sequences on its outer shell.9
In the early 1990s, toxicological assessment of C60 fullerene demonstrated that acute and chronic exposure to the molecule does not result in DNA damage and subsequent development of tumors when topically applied to mouse skin.10 Additional evidence emerged in the late 1990s that application of fullerenes to skin does not provide an appreciable allergic response in humans.11 Further studies on functionalized fullerenes also did not reveal any toxicological issues.12 It should be noted, however, that some controversy exists as to the safe and effective use of fullerenes on skin. In a 2006 article in Chemical and Engineering News, Bethany Halford challenged fullerene manufacturers and finished goods companies to provide more data and testing as to the toxicological acceptability of fullerenes in skin care preparations.13
More recent research demonstrated the utility of fullerenes in treating acne vulgaris.14 It has also been shown that fullerenes inhibit the formation of reactive oxygen species (ROS) in skin subjected to laser therapy regimens.15 Not surprisingly, there has been considerable interest in the personal care industry to develop products utilizing fullerene technology to take advantage of its therapeutic properties.16,17 Vitamin C60 BioResearch Corporation—a subsidiary of Mitsubishi Corporation and a manufacturer of fullerenes—claims that they are 172 times as effective as vitamin C.18 One should bear in mind that such a claim should take into account the details of the in vitro test for generating such data and how applicable it is to daily in vivo use. Furthermore, pro-oxidant behavior of antioxidants must not be overlooked and should be considered during efficacy and safety testing.
On the commercial side, during the 2000s, the company Zelens (London) launched Fullerene C60 Night Cream, among other products. There was also a product introduction by the Dr. Brandt line called Lineless Cream, which also used fullerenes. More recently, Vitamin C60 BioResearch Corporation appears to be working exclusively with Nova C60 Skin Solutions, who have an entire line of products based on fullerene technology.
Footnotes
1. Allotrope refers to possible crystalline forms of a molecule. For example, allotropes of carbon consist of diamond, fullerenes, and graphite.
2. H.W. Kroto, J.R. Heath, S.C. O’Brien, R.F. Curl, and R.E. Smalley, C60: Buckminsterfullerene, Nature, 318, 162-163 (1985).
3. In 1996, the Royal Swedish Academy of Sciences awarded the Nobel Prize in Chemistry to R.F. Curl, H.W. Kroto, and R.E. Smalley.
4. Z. Markovic and V. Trajkovic, Biomedical potential of the reactive oxygen species generation and quenching by fullerenes (C60), Biomaterials, 29, 3561-3573 (2008).
5. Interestingly, it has also been shown that during exposure to ultraviolet light, fullerenes undergo photosensitization reactions (transition to a long-lived excited triplet state) and react with molecular oxygen (3O2), resulting in the formation of singlet oxygen (1O2) and superoxide anion (O2•–).4 In turn, these ROS may react with a number of biological targets or even cause a cascade of molecular signaling events that could be harmful to the cell. Nevertheless, more studies are required to determine if the dual nature of fullerenes could hamper their use as free radical scavenging agents in applications involving exposure to solar radiation.
6. L. Xiao, H. Takada, K. Maeda, M. Haramoto, and N. Miwa, Antioxidant effects of water-soluble fullerene derivatives against ultraviolet ray or peroxylipid through their action of scavenging the reactive oxygen species in human skin keratinocytes, Biomed. Pharmacother., 59, 351-358 (2005).
7. Y. Saitoh, A. Miyanishi, H. Mizuno, S. Kato, H. Aoshima, K. Kokubo, and N. Miwa, Super-highly hydroxylated fullerene derivative protects human keratinocytes from UV-induced cell injuries together with the decreases in intracellular ROS generation and DNA damages, J. Photochem. Photobiol. B, 102, 69-76 (2011).
8. S. Kato, H. Aoshima, Y. Saitoh, and N. Miwa, Fullerene-C60 incorporated in liposome exerts persistent hydroxyl radical-scavenging activity and cytoprotection in UVA/B-irradiated keratinocytes, J. Nanosci. Nanotechnol., 11, 3814-3823 (2011).
9. J.G. Rouse, J. Yang, J.P. Ryman-Rasmussen, A.R. Barron, and N.A. Monteiro-Riviere, Effects of mechanical flexion on the penetration of fullerene amino acid-derivatized peptide nanoparticles through skin, Nano Lett., 7, 155–160 (2007).
10. M.A. Nelson, F.E. Domann, G.T. Bowden, S.B. Hooser, Q. Fernando, and D.E. Carter, Effects of acute and subchronic exposure of topically applied fullerene extracts on the mouse skin, Toxicol. Ind. Health, 9, 623-630 (1993).
11. A. Huczko, H. Lange, and E. Calko, Fullerenes: experimental evidence for a null risk of skin irritation and allergy, Fullerene Sci. Techn., 7, 935-939 (1999).
12. S. Kato, H. Aoshima,Y. Saitoh, and N. Miwa, Biological safety of lipofullerene composed of squalane and fullerene-C60 upon mutagenesis, photocytotoxicity, and permeability into the human skin tissue, Basic Clin. Pharmacol. Toxicol., 104, 483-487 (2009).
13. B. Halford, Fullerenes for the face: Cosmetics containing C60 nanoparticles are entering the market, even if their safety is unclear, Chem. Eng. News, 84, 47 (2006).
14. S. Inui, H. Aoshima, A. Nishiyama, and S. Itami, Improvement of acne vulgaris by topical fullerene application: unique impact on skin care, Nanomed. Nanotechnol., 7, 238–241 (2011).
15. T. Fujimoto, S. Ito, M. Ito, H. Kanazawa, and S. Yamaguchi, Induction of different reactive oxygen species in the skin during various laser therapies and their inhibition by fullerene, Lasers Surg. Med., 44, 685–694 (2012).
16. M. Lens, Use of fullerenes in cosmetics, Recent Pat. Biotechnol., 3, 118-123 (2009).
17. M. Lens, Recent progresses in application of fullerenes in cosmetics, Recent Pat. Biotechnol., 5, 67-73 (2011).
18. Fullerene, super anti-oxidant, VitaminC60 Research, In Cosmetics, August 30 (2013).