

By Gopinathan K. Menon
November 15, 2017
The Keratinocyte: A Very Popular Cell Type
As I write, a Google Scholar search turns up about 116,000 results (in 0.08 seconds) with a search for "The Keratinocyte", and since 2017, there are 6,620 results. When I drop the “Scholar” and just perform a Google search, it is 151,000. Granted, it is still not as popular as the other K search. A Google search for the Kardashians yields 58,400,000 results, but where would they be if not for their keratinocytes, which define that skin deep quality displayed in print and digital media ad nauseum? So, here is my disclaimer: I am only trying to scratch the surface of Planet Keratinocyte in a few paragraphs, describing some of the less appreciated things about this cell type that I hope will interest the cosmetic chemist.
What Is the Keratinocyte?
The keratinocyte (a cell that synthesizes keratin proteins) is the predominant cell in the epidermis (the layer of skin that overlays the dermis), which itself is divided into various strata: stratum basale (basal layer), stratum spinosum (spinous or prickly layer), stratum granulosum (granular layer), and the stratum corneum (corneous layer). Cells of the basal layer divide and move upwards, progressively differentiating (synthesizing various keratins and lipids) and passing through the other layers described above, finally ending up as cornified cells that provide the skin with a physical and chemical barrier.1,2 The outermost cells of the stratum corneum are eventually desquamated and replaced by cells below that move up in a well orchestrated fashion.3 As the keratinocytes and the nervous system share the same embryonic origin (ectoderm), keratinocytes synthesize and secrete many of the chemicals that are made by the cells of nervous system, which has given rise to the cliché, "it is our brain on the outside,"4,5 or that the epidermis is the "third brain".6
Morphology: What Does the Keratinocyte Look Like?
Depending on which epidermal layer we are examining, and what method is used to view the cell, the keratinocyte can look very different. One may employ a non-invasive technique, such as multiphoton tomography (looking from the outside in), as employed by Kohler and coworkers who described a polygonal structure for cells in the stratum corneum (horny layer), a granular cytoplasmic appearance for cells in the stratum granulosum, smaller prickle cells in the stratum spinosum, and hyperpigmented small round cells in the stratum basale.7 Conventional histology and transmission electron microscopy methods provide a more defined view of the morphological changes that keratinocytes experience as they migrate upwards through the epidermal layers, mature, and, finally, die (terminal differentiation) resulting in the formation of the stratum corneum.8
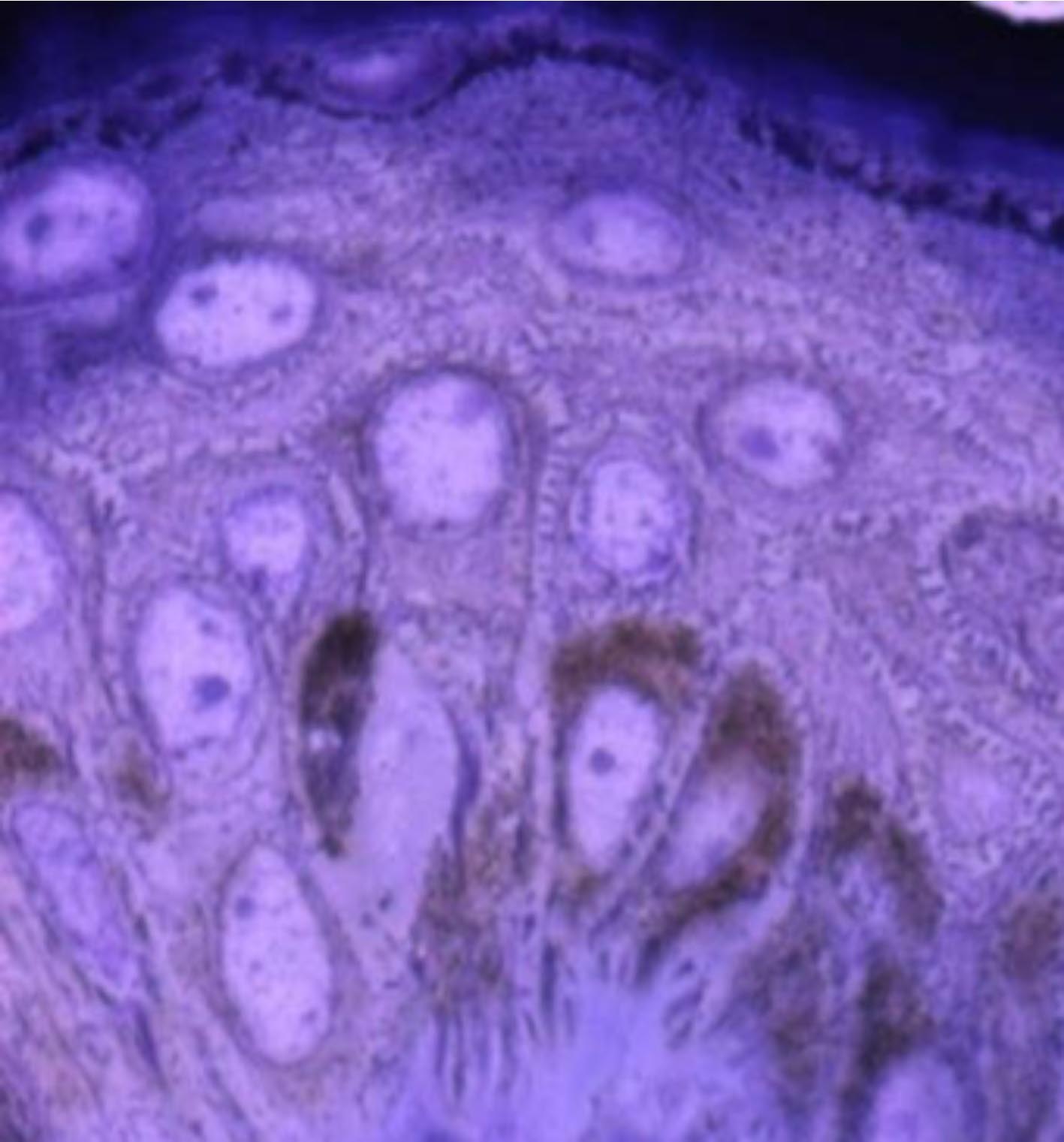
Figure 1. Histology of human epidermis shows pigment containing basal cells, spinous (prickly) cells, and granular cells marked by darkly staining keratohyalin granules. The stratum corneum lacks cellular details in histology preparations.
The Keratinocyte In Vitro
Keratinocytes can be cultured in the laboratory. Although they are not as easy to grow as fibroblasts, innumerable laboratories harvest them successfully, and employ the cultured cells for basic scientific investigations as well as potential biotechnology applications, such as the construction of artificial skin. The reconstituted 3-D skin models have a multitude of uses in skin grafting and also serve as alternative substrates for animal testing of pharmacological or cosmetic ingredients for toxicology and efficacy. Keratinocyte cultures are also employed as simple models of wound healing, for evaluating UV responses, studying carcinogenesis, and induction of cell differentiation.
Figure 2 contains a transmission electron micrograph of keratinocytes in culture. This image shows prominent nuclei as well as cytosolic organelles, such as the mitochondrion, Golgi complex, endoplasmic reticulum, and various vacuoles (multivesicular bodies and autophagic vesicles). Overall, not many keratin filaments are evident (keratin filaments appear only with initiation of keratinocyte differentiation, as normally observed when keratinocytes are exposed to higher calcium levels in the culture medium). The cellular contents of organelles may vary depending on the culture medium and/or nutritional status of the culture. Normally, cultured keratinocytes do not synthesize the epidermal lamellar bodies. However, it should be pointed out that in a skin equivalent model (3-D culture with a dermal component) lamelllar bodies are reportedly synthesized (especially with vitamin C supplementation) when keratinocytes are raised to the air-water interface to stimulate the barrier lipid production.9
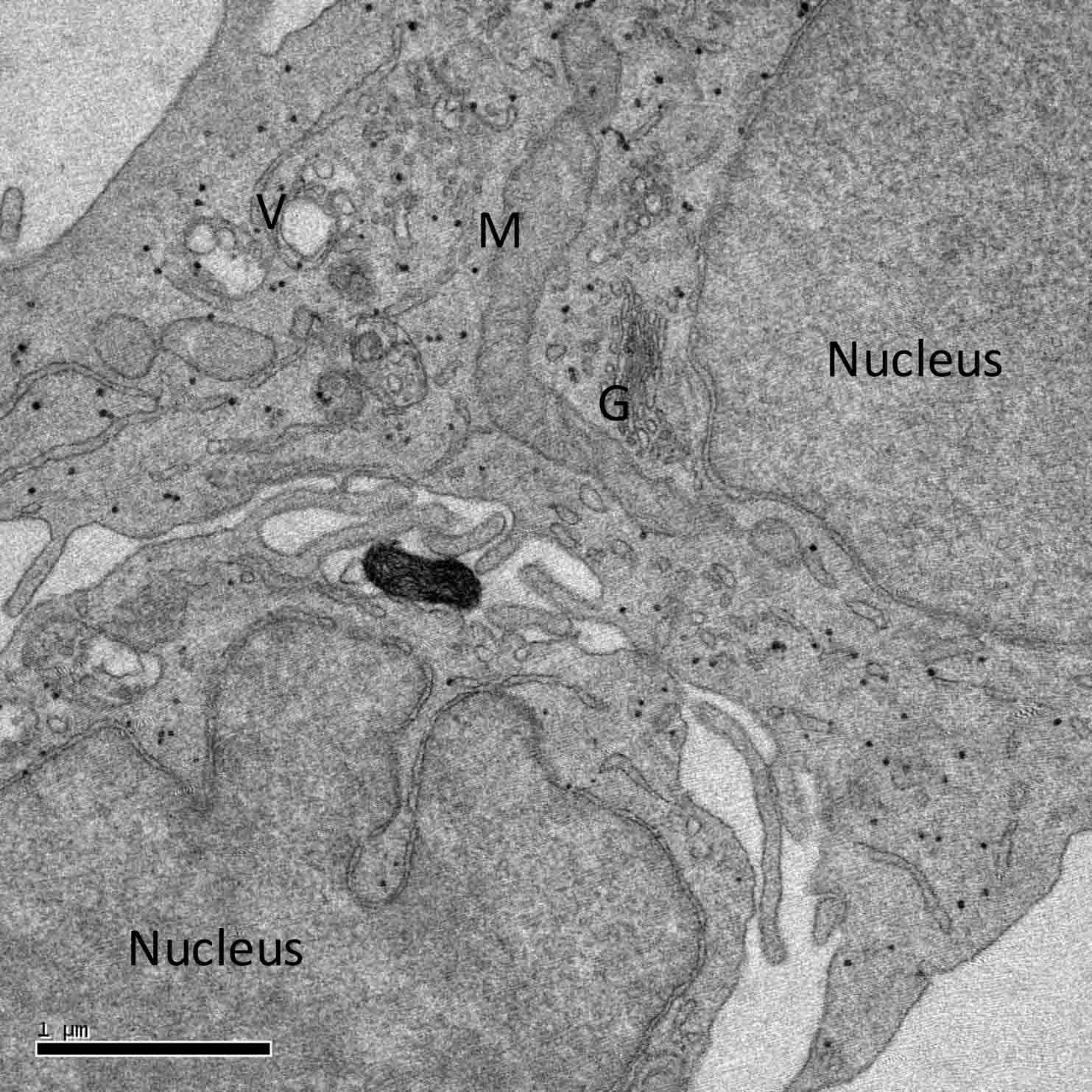
Figure 2. Transmission electron micrograph demonstrating the ultrastructure of keratinocytes in culture (non-differentiating culture conditions). Image courtesy of D. Crumrine, University of California, San Francisco.
Role of Keratinocytes in Wound Healing
It is fairly obvious to consider keratinocytes as critical to wound healing. Biophysical studies have shown that wounding of skin generates an endogenous electric field (100-200 mV/mm) in its immediate periphery. Isseroff and colleagues have shown that when keratinocytes in culture are exposed to electric currents of this magnitude, they migrate towards the cathode (i.e., the keratinocytes exhibit galvanotaxis).10 This unique behavior found in keratinocytes is in contrast to melanocytes, which did not demonstrate such a response, possibly the underlying reason for delayed re-pigmentation of healed wounds. The antimicrobial peptide (AMP) production and secretion is "AMPed Up" during such healing, playing a significant role in fighting infection when the barrier is breached.11
Cytokines and Growth Factors Secreted by Keratinocytes
Numerous cytokines, chemokines, and growth factors are secreted by keratinocytes, and these play major roles in skin sensitization, inflammation, allergic reactions, immune modulation, and neuro-endocrinologic functions of skin. Some of these parameters are useful in evaluating skin contact sensitization of low molecular weight chemicals (e.g., interleukin-18 production is a tool to discriminate contact versus respiratory allergens and/or irritants).12 Some investigators have shown that the cytokine profile of keratinocytes can be used to identify the fungal species (e.g., Arthroderma benhamiae or Trichophyton tonsurans) responsible for superficial dermatophyte infections that lead to inflammatory responses of various degrees.13 Others (Brogden lab at the University of Iowa) have exploited the differential epidermal cytokine secretory response to different stimuli as well as in aging by developing an in vivo non-invasive sampling technique and assaying cytokines from the stratum corneum that is already attracting attention from the personal care industry.14 Oxytocin, secreted by keratinocytes, could play a major role in the skin to skin contact that is integral to maternal bonding with infants.15 Does the lack of oxytocin have any possible implications for autism?16
Various Receptors Expressed in Keratinocytes
Receptors are membrane-bound cellular proteins that receive physical or chemical messages from the environment (external and internal) and cause transduction of the messages so that the cell can respond appropriately to ensure proper functioning and/or survival. There are a multitude of receptors in keratinocytes, which are positioned at the outermost surface of the cell and interface with the external environment, receiving a wide spectrum of physical and chemical stimuli. Some examples of keratinocyte receptors include the transient receptor potential channels (sensing heat, cold, etc.),17 adenosine triphosphate (ATP) receptors (ATP is the energy currency of the cell, the product of metabolism), hormone receptors (which mediate actions of hormones to modulate cell and tissue functions), and toll-like receptors (sensing infectious agents), which enable cutaneous chemosensation and contribute to information processing in the skin. It has been suggested that keratinocytes form the forefront of skin surface perception and process information to the nervous system.6 It is nearly impossible to list all of the receptors here, but a few that are novel and of recent interest are mentioned in the sections below
Cannabinoid Receptors in Keratinocytes
Epidermal keratinocytes express a functionally active mu-opiate receptor as well as its ligand, beta-endorphin, according to a report by Bigliardi-Qi and coworkers.18 They found that keratinocytes staining for beta-endorphins are clustered around unmyelinated nerve fiber terminals, which also secrete beta-endorphins, indicating direct communication between the epidermis and its nerve endings, and a mutual influence between the two cell types. The fact that keratinocytes in culture synthesize and secrete biologically active nerve growth factor (NGF) also suggests that paracrine secretion of NGF by epidermal cells influence its innervation in vivo.19 Additionally, epidermal keratinocytes release several other neuroactivators, such as calcitonin gene related peptide (CGRP), ATP, acetylcholine, glutamate, etc., in response to various stimuli. This information is evidence for complex signaling pathways between epithelial cells and sensory afferent nerve fibers.20 Disruptions in such signaling may cause (or result from) chronic skin diseases where inflammation and nerve stimulation could lead to itch and pain. Gaffal and coworkers investigated a mouse model whose keratinocyte cannabinoid receptors were deleted, and confirmed the role of cannabinoid receptors in limiting the secretion of pro-inflammatory chemokines (regulating T cell dependant inflammation) in contact allergic reactions.21
Odor Receptors in Keratinocytes
Odor receptors transmit odor signals from the nasal epithelium to the brain. Recently, they have been shown to exist in cultured keratinocytes as well as excised human skin. The functional importance of the odorant receptor OR2AT4 in keratinocytes is of interest to the cosmetic chemist, as sandalore, a synthetic sandalwood fragrance used in perfumes, was shown to be a ligand for this receptor. When this ligand binds to OR2AT4 it causes an increase in cellular calcium concentration and keratinocyte proliferation, and also cell migration in vitro, which is crucial in wound healing.22
Opsins in Keratinocytes
Opsins are photosensitive receptor proteins. The most well known of the family is rhodopsin, crucial for our eyesight. The opsin family belongs to the G protein-coupled receptors (GPCRs) superfamily. Though we routinely associate opsins with the eye, several visual and non-visual opsins, such as melanopsin and neuropsin, are also expressed in non-visual tissues such as the skin. Recently, Toh et al. described for the first time the presence of peropsin in human skin and keratinocytes, and showed that it helps detect short wavelength violet light.23 They suggest that these photoreceptors enable skin to sense and distinguish different light qualities. In the context of the use of blue light for therapeutic applications, the presence of a variety of opsins in keratinocytes holds promise for expanding the spectrum of light usage in dermatological therapy.24 These findings also provide credence for the work of Denda and colleagues who showed that different types of light have distinct effects on skin barrier repair.25
Keratinocytes in Pigmentation: Selective Role in Uptake of Melanosomes
Melanocytes are indeed responsible for the synthesis and assembly of the melanosome. The organization of the melanosomes (isolated or clustered) in keratinocytes is specific to dark or light skin types. An insightful experiment by Minwala and coworkers, heterologous co-cultures were obtained using melanocytes and keratinocytes derived from dark and light foreskin.26 In all four possible permutations, they showed that regardless of the donor melanocyte, dark skin derived keratinocytes will predominantly distribute recipient melanosomes individually, while light skin derived keratinocytes will predominantly distribute recipient melanosomes in membrane-bound clusters.
Concluding Remarks
In short, the keratinocyte is an incredibly versatile cell, which continues to fascinate us and reveal novel and unexpected properties to dedicated researchers. It is a delighting subject for those of us in quest of new biological targets for improving skin heath or enhancing its beauty.
References
1.
P.M. Elias and K.R. Feingold, Skin Barrier, CRC Press: Boca Raton, FL, 2005.
2. G.K. Menon, G.W. Cleary, and M.E. Lane, The structure and function of the stratum corneum, Int. J. Pharm., 435, 3-9 (2012).
3.
https://en.wikipedia.org/wiki/Keratinocytes.
4.
D.J. Tobin, Biochemistry of human skin—our brain on the outside, Chem. Soc. Rev., 35, 52-67 (2006).
5. M. Denda, N. Nakatani, K. Ikeyama, M. Tsutsumi, and S. Denda, Epidermal keratinocytes as the forefront of the sensory system, Exp.Dermatol., 16, 157-161 (2007).
6. M. Denda, Epidermis as the "third brain"? Dermatologica Sinica,
33, 70-73 (2015).
7.
M.J. Köhler, S. Zimmermann, S. Springer, P. Elsner, K. König, and M. Kaatz, Keratinocyte morphology of human skin evaluated by in vivo multiphoton laser tomography, Skin. Res. Technol., 17, 479-486 (2011).
8. A more detailed description of
the different layers of epidermis can be found in R.L. McMullen and G.K. Menon, Structure and function of skin: epidermis, TheCosmeticChemist.com.
9. J. Vicanová, E. Boelsma, A.M. Mommaas, J.A. Kempenaar, B. Forslind, J. Pallon, T. Egelrud, H.K. Koerten, and M. Ponec, Normalization of epidermal calcium distribution profile in reconstructed human epidermis is related to improvement of terminal differentiation and stratum corneum barrier formation, J. Invest. Dermatol., 111, 97-106 (1998).
10. R.K. Sivamani, M.S. Garcia, and R.R. Isseroff,
Wound re-epithelialization: modulating keratinocyte migration in wound healing, Front. Biosci., 12, 2849-2868 (2007).
11. Y. Lai and R.L. Gallo, AMPed Up immunity: how antimicrobial peptides have multiple roles in immune defense, Trends Immunol., 30, 131-141 (2009).
12. E. Corsini, V. Galbiati, M. Mitjans, C.L. Galli, and M. Marinovich, NCTC 2544 and IL-18 production: a tool for the identification of contact allergens, Toxicol. In Vitro, 27, 1127-1134 (2013).
13. Y. Shiraki, Y. Ishibashi, M. Hiruma, A. Nishikawa, and S. Ikeda,
Cytokine secretion profiles of human keratinocytes during Trichophyton tonsurans and Arthroderma benhamiae infections, J. Med. Microbiol., 55(Pt 9), 1175-1185 (2006).
14. P.M. Kinn, G.O. Holdren, B.A. Westermeyer, M. Abuissa, C.L. Fischer, J.A. Fairley, K.A. Brogden, and N.K. Brogden, Age-dependent variation in cytokines, chemokines, and biologic analytes rinsed from the surface of healthy human skin, Sci. Rep., 5, 10472 (2015).
15. S. Denda, K. Takei, J. Kumamoto, M. Goto, M. Tsutsumi, and M. Denda,
Oxytocin is expressed in epidermal keratinocytes and released upon stimulation with adenosine 5'-[gamma-thio]triphosphate in vitro, Exp. Dermatol., 21, 535-537 (2012).
16. E. Hollander, J. Bartz, W. Chaplin, A. Phillips, J. Sumner, L. Soorya, E. Anagnostou, and S. Wasserman, Oxytocin increases retention of social cognition in autism, Biol. Psychiatry, 61, 498-503 (2007).
17. M. Denda and M. Tsutsumi, Roles of transient receptor potential proteins (TRPs) in epidermal keratinocytes, Adv. Exp. Med. Biol., 704, 847-860 (2011).
18. M. Bigliardi-Qi, L.T. Sumanovski, S. Büchner, T. Rufli, and P.L. Bigliardi, Mu-opiate receptor and beta-endorphin expression in nerve endings and keratinocytes in human skin, Dermatology, 209, 183-189 (2004).
19. E. Di Marco, P.C. Marchisio, S. Bondanza, A.T. Franzi, R. Cancedda, and M. De Luca, Growth-regulated synthesis and secretion of biologically active nerve growth factor by human keratinocytes, J. Biol. Chem., 266, 21718-21722 (1991).
20. M. Ritter-Jones, S. Najjar, and K.M. Albers, Keratinocytes as modulators of sensory afferent firing, Pain, 157, 786-787 (2016).
21. E. Gaffal, M. Cron, N. Glodde, T. Bald, R. Kuner, A. Zimmer, B. Lutz, and T. Tüting, Cannabinoid 1 receptors in keratinocytes modulate proinflammatory chemokine secretion and attenuate contact allergic inflammation, J. Immunol., 190, 4929-4936 (2013).
22. D. Busse, P. Kudella, N.M. Grüning, G. Gisselmann, S. Ständer, T. Luger, F. Jacobsen, L. Steinsträßer, R. Paus, P. Gkogkolou, M. Böhm, H. Hatt, and H. Benecke,
A synthetic sandalwood odorant induces wound-healing processes in human keratinocytes via the olfactory receptor OR2AT4, J. Invest. Dermatol., 134, 2823-2832 (2014).
23. P.P. Toh, M. Bigliardi-Qi, A.M. Yap, G. Sriram, and P. Bigliardi,
Expression of peropsin in human skin is related to phototransduction of violet light in keratinocytes, Exp. Dermatol., 25, 1002-1005 (2016).
24. I. Castellano Pellicena, N.E. Uzunbajakava, C. Delatorre, and J. Thornton, 115 Opsins and cryptochromes in human epidermal keratinocytes: a perspective for blue light therapies, J. Invest. Dermatol., 137, S212 (2017).
25. M. Denda and S. Fuziwara,
Visible radiation affects epidermal permeability barrier recovery: selective effects of red and blue light, J. Invest. Dermatol., 128, 1335-1336 (2008).
26. L. Minwalla, Y. Zhao, I.C. Le Poole, R.R. Wickett, and R.E. Boissy,
Keratinocytes play a role in regulating distribution patterns of recipient melanosomes in vitro, J. Invest. Dermatol., 117, 341-347 (2001).