

by Gertrude-Emilia Costin, Ph.D., M.B.A.
Institute for In Vitro Sciences, Inc. (IIVS)
30 W Watkins Mill Road, Suite 100
Gaithersburg, MD, United States
November 15, 2016
The melanocyte “residence” in the basal layer of the epidermis.
Whether admiring a majestic butterfly on a field of flowers, being puzzled by the incredible flexibility of a chameleon to switch the “color gears”, or being attracted to the patterns of a tiger or the joyful appearance of a parrot (Figure 1), one time or another we must have been fascinated by the colorful show nature offers. And at least once, we probably wondered in a scientific way, “Who is responsible for it?” The immediate answer is: a pigment identified as melanin. Still, there is a biologic player “hard at work” producing the pigments that generate great variations in human skin color and is the protagonist of this article. It is the melanocyte. While the melanocyte often comes across as a “celebrity” due to the attention it has been given by medical doctors and researchers alike—and to its incredible plasticity and capability to manipulate appearance—there is much more to its activity than the easily visible variation in color that meets the eye. The melanocyte is a master of communication, a molecular trait given by its “ancestors”, the neural crests it derives from. Therefore, this unique cell should be addressed by integrating it in its physiological environment and deciphering its relationship with other “residents” of skin.

Figure 1:
The color palette in nature.
The great diversity in the color of human skin. Pigmentary skin conditions.
The constitutive color of human skin varies widely across the globe, from the very pale as in Celtic skin to the very dark present in regions such as sub-Saharan Africa. Skin color is frequently classified into one of six distinct skin phototypes.1 This is the most commonly used method to categorize skin color depending on its ability to tan/burn following exposure to ultraviolet (UV) radiation (Figures 2a-2b). The color is generated by the melanin produced in melanocytes and distributed throughout the skin following a highly regulated complex pathway and equally sensitive to any factors that could destabilize the biochemical and biological equilibrium in the skin.
Across the array of skin colors, numerous pigmentary conditions affect humans from the most devastating such as oculocutaneous albinism (Figure 2c) or vitiligo (Figure 2d) to melasma present during pregnancy (Figure 2e), post-inflammatory hyperpigmentation (Figure 2f), or lentigines (Figure 2g), which all have a significant psychosocial impact on the affected individuals.
Albinism is a heterogeneous group of genetic hypopigmentary disorders clinically divided into ocular albinism and oculocutaneous albinism (Figure 2c).2 All types of oculocutaneous albinism discovered thus far are associated with reduced/absent pigmentation of the skin, hair, and eyes, which occurs as a consequence of mutations in genes involved in the regulation of melanin biosynthesis.3-10 Vitiligo is the most common acquired type of leukoderma, which is induced as a result of epidermal melanocyte destruction11-12 that is caused by immune attack by melanocyte specific T cells, innate immunity, and melanocyte-specific antibodies.13-14 The hallmark of the disease is white patches of the skin (Fig. 2d).
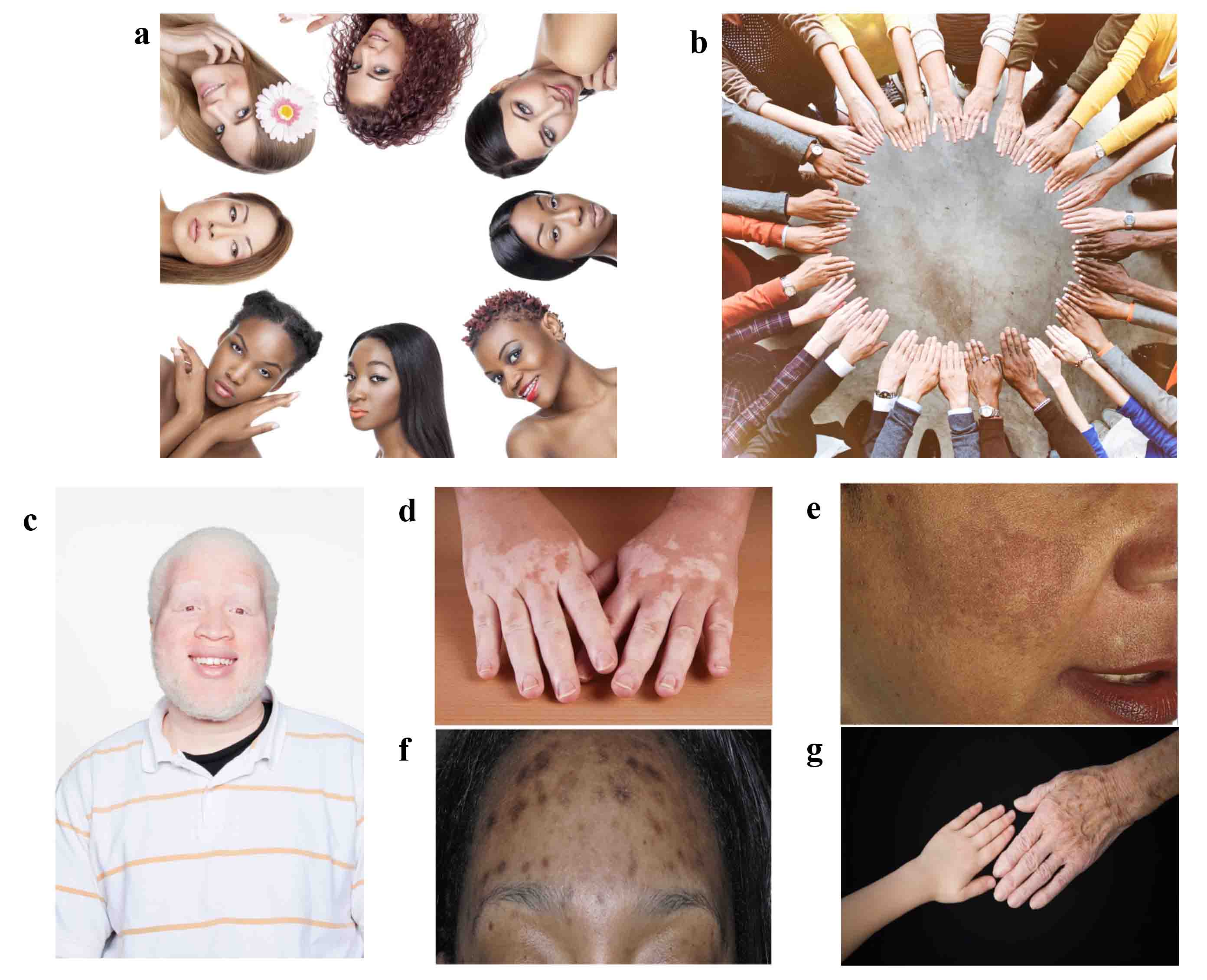
Figure 2:
The variation of human skin color (a-b) and pigmentary conditions: oculocutaneous albinism (c); vitiligo (d); melasma of pregnancy (e); post-inflammatory hyperpigmentation (f); lentigines (g). Panels (e-f) reproduced with permission from C.N. Lawson et al., Updates in the understanding and treatments of skin & hair disorders in women of color, Int. J. Wom. Dermatol., 1, 59-75; copyright (2015) Elsevier.
Melasma (chloasma) is an acquired pigmentary disorder characterized by altered cutaneous pigmentation associated with pregnancy that occurs in women of all racial and ethnic groups.15 Besides the elevated levels of sex hormones in pregnancy, other factors responsible for this condition include genetic predisposition and UV light exposure.16 Post-inflammatory hyperpigmentation is an acquired hypermelanosis occurring in all skin types, with a higher incidence in skin-of-color patients (Fitzpatrick skin types IV through VI) (Figure 2f).17 Post-inflammatory hyperpigmentation represents a pathophysiological response to cutaneous inflammation, acne lesions, ingrown hairs, scratches, insect bites, atopic dermatitis, lichen planus, chronic dermatitis, and phototoxic eruptions (allergic reactions to products coupled with exposure to sunlight).18-22 The lentigines (age spots or liver spots) are hyperpigmented spots (Figure 2g) typical in photoaging—usually induced by exposure to UV light and the resultant chronic inflammation.23 Lentigines occur on skin areas prone to sun exposure, such as hands, arms, and face.
Regardless of the nature of the pigmentary condition, the general desire of affected individuals is to achieve uniformity of skin color. Doctors and researchers have been equally challenged to address these skin conditions by designing either medical treatments or cosmetic products to resolve or ameliorate their gravity. In order to be able to design efficacious treatment regimens, there is an unavoidable need to go “skin deep” and investigate the melanocyte in its environment, understand the factors affecting its activity, and attempt to reestablish the physiological equilibrium that is ideal for the melanocyte functionality.
Melanocyte Residence and Activity
While melanocytes are found throughout the human body—from the brain to the lungs and beyond (Figure 3)—their main residence remains the skin, in the basal layer of the epidermis, where they play multiple roles. The three most visible traits of human race, skin, hair and eye color, depend on the amount, size, and type of melanins produced by melanocytes, and also on the subsequent transfer of the pigment to the keratinocytes.24 Melanin plays a critical defense role against the harmful effects of UV radiation due to its ability to absorb and reflect UV energy and to scavenge oxidative free radicals.25
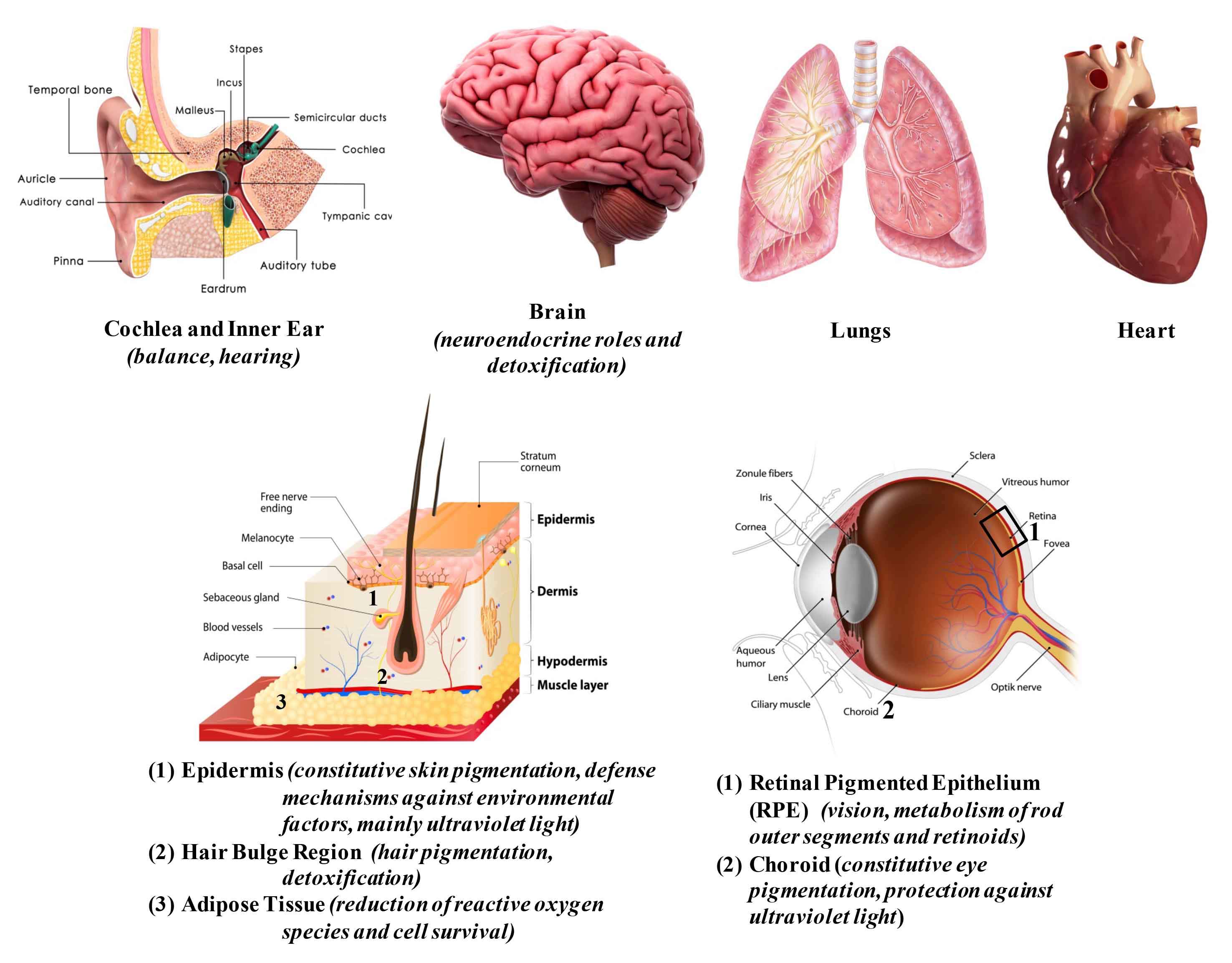
Figure 3: Distribution of melanocytes in human tissues.
Melanin is produced within highly specialized organelles named melanosomes, which transition through four stages during their development. The pre-melanosomes (Stage I) present as round, small vesicles with an amorphous matrix (Figure 4a). Stage II melanosomes exhibit a structured fibrillar matrix; at this stage, tyrosinase—the principle enzyme of the melanogenic pathway—is present, but pigment synthesis is not generally observed. The beginning of melanin production takes place in Stage III and the pigment is deposited on protein fibrils. In the last stage of their development (Stage IV), the melanosomes are fully melanized (Figure 4a), lose the tyrosinase activity, and are transported to surrounding keratinocytes by elements of the cytoskeletal system. Through their dendrites, the melanocytes establish close contacts with their "best friends", the neighboring keratinocytes. Together they form the “epidermal melanin unit” usually composed of 1 melanocyte in contact with 35-40 neighboring keratinocytes, and work together as a team to produce and distribute melanin in the skin. As already inferred, the melanocyte may be the center piece in the puzzle defining color; however, by its communicative nature it is a highly “socializing” cell that collaborates with others present in the skin, such as Langerhans, fibroblast, and endothelial cells. All these cell types compose the functional unit in charge of regulating the melanocyte function and phenotype of the skin through secreted factors and their receptors and via cell-to-cell contacts.26-28
Figure 4: Positioning the melanocyte in its physiological environment.
Melanogenic Pathway
There are two types of melanin in the human skin: the black/brown eumelanins and red/yellow pheomelanins, which exhibit distinct physical and biological properties.29,30 Eumelanin has a higher resistance to degradation and ability to neutralize reactive oxygen species (ROS) compared to pheomelanin, and therefore is attributed better photoprotecting properties. Despite the wide diversity observed in human race (Figures 2a and 2b), the density of melanocytes in all types of skin is virtually identical31,32 and the differences in skin color depend on the amount of melanin produced, the efficiency of its transfer from melanocytes to keratinocytes, and the ratio of pheomelanin to eumelanin synthesized.33-36
The types of melanins synthesized depend on the availability of substrates and the specific function of the melanogenic enzymes involved in the process (Figure 4b).37 Tyrosinase is the rate limiting enzyme of the pathway and catalyzes the first two steps: the hydroxylation of tyrosine to L-3,4-dihydroxyphenylalanine (DOPA) and its rapid oxidation to DOPAquinone. When available, DOPAquinone interacts with cysteine producing 3- or 5-cysteinylDOPAs (CDs), which then oxidize, polymerize, and generate the pheomelanin. In the absence of thiols (cysteine, glutathione, or thioredoxin), brown-black eumelanin is produced. DOPAquinone spontaneously undergoes cyclization to DOPAchrome, which then loses carboxylic acid and generates 5,6-dihydroxyindole. In turn, 5,6-dihydroxyindole oxidizes rapidly and polymerizes to form the dark brown-black, insoluble melanin. However, in the presence of another key enzyme of the pathway, DOPAchrome tautomerase, DOPAchrome will form 5,6-dihydroxyindole-2-carboxylic acid. Tyrosinase and tyrosinase-related protein 1 catalyze further conversions that finally produce the lighter brown color melanin based on 5,6-dihydroxyindole (Figure 4b).
In reality, melanocytes produce mixtures of eumelanin and pheomelanin, qualifying the biosynthesis pathway to a three-step pathway—mixed melanogenesis—based on available substrate(s).38 The first stage is the production of cysteinylDOPA isomers which takes place as long as the cysteine concentration is kept above 0.13 μM. The second stage is represented by the oxidation of 3- or 5-cysteinylDOPA to produce pheomelanin, a process that continues as long as 3- or 5-cysteinylDOPA is present at concentrations above 9 μM. The last stage is the production of eumelanin, which can begin only after the majority of 3- or 5-cysteinylDOPA and cysteine is depleted. Therefore, the ratio of eumelanin to pheomelanin is determined by tyrosinase activity and the availability of key substrates (tyrosine and cysteine) in the melanosomes.39
Regulation of the Melanogenic Pathway
I. By Extrinsic Factors
The complexity of the melanogenic pathway warrants a highly regulated interplay of multiple factors acting cohesively to keep the process working within physiological parameters. Melanocytes are highly responsive cells to their environment and modulate their levels of melanin production or proliferation according to extrinsic signals (such as UV radiation), or intrinsic factors derived from other cell types in the skin.28,40,41 UV radiation can act directly on melanocytes or influence their activity by signaling through factors produced by keratinocytes. Within melanocytes, UV light primarily stimulates the expression and function of the key enzymes involved in the melanogenic pathway (tyrosinase, tyrosinase-related protein 1, and DOPAchrome tautomerase), but its action is not limited to these enzymes.35,42 Exposure to UV radiation up-regulates the expression of the oncogene, p53, in keratinocytes, which directly stimulates the production of multiple factors that can then act in a paracrine manner to stimulate melanocyte function through receptor-mediated signaling pathways (Figure 4c). Exposure of skin to UV radiation facilitates the distribution of melanin to the supranuclear areas of the keratinocytes to protect them against DNA damage, and to the upper epidermis to protect the lower epidermis, which hosts melanocyte and keratinocyte stem cells.43
II. By Intrinsic Factors Synthesized within Melanocytes
Besides the enzymatic battery responsible for melanin production (tyrosinase, tyrosinase-related protein 1, and DOPAchrome tautomerase), many other proteins have been determined to play supportive roles in the process, such as ocular albinism 1 protein, P protein, membrane-associated transporter protein, copper-transporting P-type ATPase, and biogenesis of lysosome-related organelles complex 1.44 Equally critical is the complex of structural components within melanosomes, represented by Pmel17/gp100/Silver protein, whose sorting involves adaptor protein 1A, adaptor protein 1B, adaptor protein 2, and spectrin, and by the chaperone-like component melanoma antigen recognized by T-cells 1, which plays structural roles in melanosome maturation.45-47 Multiple factors are involved in the melanosome movement and transfer to keratinocytes, such as microtubules, dynein, kinesin, actin filaments, Rab27a, melanophilin, myosin Va, and Slp2-a (Figure 4d). Other critical molecular players that affect skin pigmentation belong to the transcription factors family and include cyclic Adenosine monophosphate response element binding protein, microphthalmia-associated transcription factor, paired box 3, SRY-Box 9/10, and T-cell factor/lymphoid enhancer factor.
III. By Intrinsic Factors Synthesized by other Cells Residing in the Skin
The cross-talking between different signaling pathways within the skin also involves fibroblasts, endothelial cells, and inflammatory cells that are part of an epidermal complex network responsible for the maintenance of skin homeostasis (Figure 4e). For example, among the factors secreted by dermal fibroblasts are stem cell factor, neuregulin 1,48 or Dickkopf 1,49-51 which influence the growth and pigmentation of melanocytes, their shape, dendricity, mobility, and adhesive properties.52 Endothelial cells produce endothelin, prostaglandins, and nitric oxide. For example, nitric oxide is capable to initiate melanogenesis, erythema, and immunosuppression in response to UV radiation (and can be also secreted by inflammatory cells). Hormonal factors that stimulate pigmentation include estrogen (which is related to pregnancy-induced pigmentation), α-melanocyte stimulating factor (MSH), adrenocorticotropic hormone, and endorphin (Figure 4e), while androgens have inhibitory effects on melanocytes.53 Arachidonate-derived chemical mediators, especially prostaglandins (PGE2 and PGF2α), leukotrienes (LTC4 and LTD4), and thromboxanes (TXB2) are secreted by inflammatory cells and enhance tyrosinase activity and subsequently the melanin synthesis.52 Furthermore, pro-inflammatory cytokines [interleukin (IL)-1α, IL-2, IL-3, IL-6, IL-10 and tumor necrosis factor (TNF)-α] and chemokines (IL-8, C-C Motif Chemokine Ligand 2) can modulate the melanocyte activity; of these, IL-1, IL-6, and TNF-α inhibit melanogenesis.54 The other factors produced by inflammatory cells stimulate the production of melanin (catecholamines, eicosanoids, serotonin, α-MSH, and nitric oxide) by acting in an autocrine manner.55,56 Finally, since melanocytes and neurons are derived from neural crest cells during embryogenesis, nerve cell-derived factors including neurotrophins (neuronal growth factor) and neuropeptides (calcitonin gene-related peptide) have stimulatory effects on melanocytes, thus supporting the neuronal theory to explain the occurrence of segmental vitiligo.57
Given the complexity of the pathways involved in melanin synthesis and the regulatory mechanisms, the opportunities to influence the production of pigment(s) are numerous and tempting and minute changes can induce extreme effects in the appearance of skin. In the following sections, strategies to design such modulators particularly for cosmetic purpose will be discussed along with laboratory tools to test their efficacy.
The common strategies used to design safe and efficacious modulators of melanogenesis follow the intracellular processing of TYR, the main enzyme of melanin biosynthesis, as it undergoes transcription and maturation, is transported to melanosomes, and eventually degraded (Figure 4f). These strategies aim not only to inhibit melanin formation de novo, but also to lower hyperpigmentation induced by various factors acting on skin.
In general, tyrosinase-related proteins (tyrosinase, tyrosinase-related protein 1, and DOPAchrome tautomerase) are localized in the melanosomal membrane58 and share several structural properties: a cytoplasmic and transmembrane domain, a glycosylated luminal domain including the cysteine-rich epidermal growth factor motif, and two metal-binding regions involved in the structure of the catalytic site.59 Tyrosinase is a single chain glycoprotein specifically synthesized within melanocytes, where it transits through the endoplasmic reticulum and Golgi network, and is finally transported to melanosomes.60 The enzymatic activity of tyrosinase is dependent on the presence of two copper binding sites where three histidine residues coordinate the copper atom at each site, and both copper atoms coordinate an O2 molecule.61 From a total of six potential glycosylation sites, three are located within or in close proximity to the active site domains.59
The ideal depigmenting compound should meet several criteria: be safe, particularly with repeated use; have a potent, rapid, and selective depigmenting effect on stimulated melanocytes; carry no short- or long-term side-effects; lead to permanent removal of undesired pigment; and impact one or more steps of the pigmentation process if at all possible.
Despite the large number of tyrosinase inhibitors demonstrated to have skin lightening effects in vitro, only a few are actually able to induce effects in clinical trials. Possible challenges regarding the efficacy of skin color modulators are: the stability of the finished products; their toxicity; and the skin penetration rate, which can be limited by the stratum corneum thereby preventing tyrosinase inhibitors from reaching the desired target. When transitioning from efficacy studies conducted in vitro to clinical trials, combinations of two or more agents acting on different mechanisms should be evaluated in order to achieve synergistic effects.62,63 This strategy may eventually allow application of lower concentrations of each agent, thus possibly reducing adverse effects and improving the expected outcome of the clinical results.
The number of ingredients that have inhibitory effects on the melanogenic pathway is impressive (Table 1), while there are not as many demonstrating stimulatory effects on melanin synthesis (Table 2). The reasons can be many and among those worth mentioning are: the availability of said ingredients; the interest and need expressed by the consumers and patients; and the apparently high feasibility to reduce pigmentation compared to increasing it. In the author’s opinion, the most popular ingredients seem to be the inhibitors of tyrosinase enzymatic activity followed by glycosylation inhibitors and agents impacting the melanin transfer to keratinocytes (Table 1). Some of the ingredients affecting tyrosinase enzymatic activity have been subject to extensive research, which in time demonstrated their adverse effects (e.g., hydroquinone; see Table 1 and associated notes) or instability when formulated. From the group of glycosylation inhibitors, an interesting example is N-butyldeoxynojirimycin, an inhibitor of the endoplasmic reticulum-processing enzymes α-glucosidases I and II, initially patented for Gaucher disease.64 An in vitro study using mouse melanoma cells treated with N-butyldeoxynojirimycin indicated that glycosylated immature N-glycans do not prevent the transport of tyrosinase to its specific cellular organelle, but that its conformation may be altered, resulting in a significant reduction of its enzymatic activity.65
Newer generations of melanogenic modulators concentrate on little or unexplored territories, such as innovative approaches to impact established steps in the melanin processing (e.g., melanin transfer to keratinocytes), or chemical classes of ingredients never investigated for their impact on melanin biosynthesis pathway, or tyrosinase processing and beyond. For example, a recent study by Diwakar et al. demonstrated for the first time that Neu5Acα(2-6)Gal- and possibly sialyl(α2-3)gal-terminated glycans play key roles in melanin synthesis and melanosome transfer to keratinocytes.66 These structures were prominently expressed on melanocyte dendrites where they are closely associated with the transfer of melanosomes to keratinocytes, a rate limiting step in skin pigmentation (Figures 4f, Step 4). These findings point to new possibilities for reducing hyperpigmentation through inhibition of the synthesis and function of oligosaccharides regulating the pigmentary system.
In recent years, investigators have turned their attention to short-sequence peptides as potential therapeutic candidates for the treatment of skin disorders. The most widely studied and popular signal peptide is the sequence lysine–threonine–threonine–lysine–serine found in type-I procollagen.67 This pentapeptide has been shown to stimulate feed-back regulation of new collagen synthesis and to significantly improve the appearance of photoaged human facial skin.68,69 A study by Abu Ubeid et al. screened an oligopeptide library and discovered several short-sequence peptides that showed promising results when tested in vitro.70
A recent puzzling report demonstrated that physiologic estrogen (17β-estradiol) and progesterone reciprocally regulate melanin synthesis.71 The results are intriguing because primary human melanocytes are known to lack classical estrogen or progesterone receptors. The authors demonstrated that the effects of sex steroids on human pigment synthesis are mediated by the membrane-bound, steroid hormone receptors: G protein-coupled estrogen receptor as well as progestin and adipoQ receptor 7. Further investigations into the matter may be able to identify additional therapeutically useful receptors in melanocytes and help clarify how cumulative GPCR signaling is involved in the regulation of melanin production in human skin.
The discrepancy between the results generated in vitro and in clinical studies suggests that innovative laboratory tools and strategies are needed for validating the efficacy and safety of actives with potential skin lightening effects. Some of these possibilities are discussed in the following section.
The previous sections of this article detail the role of the melanocyte and other cell types working cohesively to modulate the fine-tuned regulatory mechanisms responsible for skin color. There are multiple possibilities to impact the melanogenic pathway and, over the years, the advances in science have provided more complex, sophisticated, and relevant laboratory tools and strategies to assist in the design of efficacious agents that can either decrease or increase the melanin production. By going “skin deep”, from the visible effects at the tissue level to the very deep level of the melanosome, or even purified enzymes of the melanogenic pathway, many intimate interactions between cells and molecular factors have been and continue to be discovered (Figure 5, left side, arrow pointing down). However, if this journey into the depth of the skin is thought of as a trip by a funicular, the journey back to the surface is needed to address skin pigmentary conditions and can be completed by using relevant laboratory tools investigating the efficacy of actives capable to modulate skin color.
Figure 5: Going “skin deep” and the journey back to the skin surface: available assays and testing strategies integrated into the skin care products manufacturing framework.
In industry, the path to carry an idea/concept to completion (the finished product available to the consumer) can take various “side roads” (Figure 5). Nowadays, the general approach starts with casting a wide net to investigate potential ingredients with melanogenic action by using modeling or docking approaches. The transition towards bench type experiments starts from the deepest level within the skin due to the ease to conduct such projects, from organelles to cells to complex tissue models (most recently reviewed in Gunia-Krzyazak and coworkers), and finally reaching the clinical trial stage.72 In the process, the cosmetic industry needs to transition from the proof of principle stage (often addressed using in vitro assays) to safety and efficacy testing (which can occur sequentially in either order or concurrently) to demonstrating action in clinical studies, and finally to launching a product on the market.
To start the “journey to surface” (Figure 5, right side, arrow pointing up), modeling can be employed by using for example the Shape Signatures, a novel virtual approach based on a computational algorithm for rapid screening of chemical libraries. This method converts the size and shape of a molecule, as well as its surface charge distribution and other bio-relevant properties, into compact histograms (signatures) that allow for rapid comparison between molecules.73 Using this approach, Ai and coworders identified a novel class of depigmentation agents that demonstrated promise for skin lightening product development and that were experimentally confirmed as potent tyrosinase inhibitors.74,75
The catalog of in vitro assays available nowadays is diverse and offers testing possibilities from the simplest to the very complex. An initial evaluation of the depigmenting properties of various agents can be performed on purified tyrosinase. Mushroom source tyrosinase is the classic choice in many laboratories, although mammalian-derived tyrosinase should be considered as more relevant. Other melanogenic enzymes may be useful targets for these assays that could provide rapid screening of novel agents, promoting them as candidates for the next phase. These methods allow for the screening of a potentially large numbers of compounds for their inhibitory or stimulatory effects on the specific processes being evaluated, are relatively inexpensive, and can be readily performed in a 96-well plate, allowing one to conduct rapid robotic high-throughput experiments.76 At this stage, the number of candidates revealed by the modeling approach could be decreased by confirmation in the enzymatic assay, thus promoting the most promising molecules within the group to the next set of available assays.
The next step would be to use a test system with greater complexity, such as melanocyte cultures that can be designed in a multi-well plate format for moderate throughput screening of the cytotoxicity and effects on melanin synthesis. For more in-depth biological evaluation, melanocyte/keratinocyte co-culture systems and pigmented reconstructed skin models seem to be reliable methods to screen the capability of new agents to interfere with the pigmentary process, in particular following triggering stimuli such as UV irradiation or exposure to other agents.
The co-culture systems represent a step forward from the simple melanocyte cultures since some of the physiological interactions with the keratinocytes can be addressed, including the melanosome transfer between the two cell entities. For example, such co-culture model systems have been designed by using immortalized77 or primary melanocytes78 in association with keratinocytes. These test systems are still challenged by limited spatial interactions and by the solubility of agents tested as most of the cosmetic industry final formulations are creams or lotions. Furthermore, with each transition from a simple to a more complex assay, some discrepancies in results can be eventually resolved. For example, arbutin is a more potent inhibitor of melanin synthesis on melanocytes co-cultured with keratinocytes than on pure melanocyte cultures, demonstrating the advantage of mixed culture systems that include cell-cell interactions, similar to that found in native human skin.79
Many of the issues encountered with cell cultures and co-cultures have been overcome by using reconstructed pigmented skin models commercially available from various companies: MelanoDerm™ from MatTek Corporation (Ashland, MA, USA);80-82 reconstructed Human Pigmented Epidermis from EpiSkin (Lyon, France);83 epics-M from Cell Systems (Troisdorf, Germany); and NeoDerm-ME from TegoScience (Seoul, Korea). The possibility to design pigmented reconstructed tissue models that reflect the various skin phototypes is a clear advantage, especially for mechanistic studies. These models also provide a more physiologically relevant system to study the 3-dimensional interactions between melanocytes and keratinocytes, and to elucidate the regulatory mechanisms of melanogenic compounds. As with any in vitro test system, there are several limitations the reconstructed skin models have: their structure begins to deteriorate after 3 weeks, preventing long-term studies and presenting challenges due to the ongoing differentiation of the model in culture, thereby making it difficult for the actives to reach the basal layer where the melanocytes reside. The manufacturers always aim to improve their models, and one step forward would certainly be the inclusion of fibroblasts or other relevant cell lineages described earlier in this article into the models to allow more complex cellular and molecular interactions to occur and to reveal action of raw ingredients or final formulations to modulate skin color in vitro.
Overall, the large number of reports publicly available demonstrate that the simpler and less expensive test systems are quite useful for routine screening of melanogenic modifiers. However, bioactives with promising potential should be tested on pigmented reconstructed skin models before eventually launching a clinical trial. The design of the multiple in vitro assays described thus far originate from the need to replace animal testing due to social and political pressure, and to address regulations in the cosmetic industry not allowing such testing for ingredients and finished products.84
Other available assays use imaging techniques that quantify melanin in three-dimensional skin models and in skin in situ.42,85 A non-invasive tool—diffuse reflectance spectroscopy—was recently used to predict human sensitivity to UV exposure. Melanin content was measured using diffuse reflectance spectroscopy and was compared to the densitometric analysis of Fontana Masson staining in skin biopsies obtained from subjects (Asian, African American, and Caucasian) one day after exposure to one minimal erythemal dose (MED) of UV. The use of diffuse reflectance spectroscopy alone proved to be a predictive model for the non-invasive determination of UV sensitivity in humans that should be invaluable for determining appropriate UVR doses for therapeutic, diagnostic, and/or cosmetic devices.
Finally, when conducting clinical trials, the activity of the hypopigmenting formulation is assessed using non-invasive techniques (Figure 5), such as remittance spectrophotometry and UV light photography to obtain comparable values, or more advanced imaging methods capable to specifically measure the melanin chromophore.86-89
The cosmetic industry finds itself in a continuous quest to design safe, effective and novel ingredients and products that can address multiple pigmentary skin conditions. Scientists from industry and academia join forces to standardize and improve the lab tools and techniques that can provide reliable, fast, and relevant results about the safety and efficacy of the promising prototypes. The need for novelty and innovation motivates industry to go beyond the much investigated action of ingredients on the key melanogenic enzyme, tyrosinase. Learning from heavily investigated clinical skin pigmentary conditions, such as vitiligo or melanoma, researchers have the opportunity to concentrate on other enzymes and factors involved in the melanogenic pathway. By investigating multiple steps within the melanogenic pathway and beyond, one may find the key to the design of breakthrough products able to improve the consumer’s health, social life, and psychological well being.
1. T.B. Fitzpatrick, The validity and practicability of sun-reactive skin types I through VI, Arch. Dermatol., 124, 869-871 (1988).
2. R.A. King, W.S. Oetting, C.G. Summers, D.J. Creel, and V.J. Hearing, "Abnormalities of pigmentatio" in Emery and Rimoin’s Principles and Practice of Medical Genetics, Eds. D.L. Rimoin, J.M. Connor, R.E. Pyeritz, and B.R. Korf, Churchill Livingstone: New York (2006).
3. R.A. Spritz, K.M. Strunk, L.B. Giebel, and R.A. King, Detection of mutations in the tyrosinase gene in a patient with Type IA oculocutaneous albinism, New Eng. J. Med., 322, 1724-1728 (1990).
4. R.E. Boissy, H. Zhao, W.S. Oetting, L.M. Austin, S.C. Wildenberg, Y.L. Boissy, Y. Zhao, R.A. Sturm, V.J. Hearing, R.A. King, and J.J. Nordlund, Mutation in and lack of expression of tyrosinase related protein 1 (TRP1) in melanocytes from an individual with brown oculocutaneous albinism: a new subtype of albinism classified as OCA3, Am. J. Hum. Gen., 58, 1145-1156 (1996).
5. P. Manga, J.G. Kromberg, N.F. Box, R.A. Sturm, T. Jenkins, and M. Ramsay, Rufous oculocutaneous albinism in southern African Blacks is caused by mutations in the TYRP1 gene, Am. J. Hum. Gen., 61, 1095-1101 (1997).
6. P. Manga P. and S.J. Orlow, The pink-eyed dilution gene and the molecular pathogenesis of tyrosinase-positive albinism (OCA2), J. Dermatol., 26, 738-747 (1999).
7. R.A. King, "Albinism" in The Pigmentary System: Physiology and Pathophysiology, Eds. J.J. Nordlund, R.E. Boissy, V.J. Hearing, R.A. King, and J.P. Ortonne), Oxford University Press: New York (1998).
8. W.S. Oetting and R.A. King, Molecular basis of albinism: mutations and polymorphisms of pigmentation genes associated with albinism, Hum. Mutat., 13, 99-115 (1999).
9. L.L. Baxter and W.J. Pavan, The oculocutaneous albinism type IV gene Matp is a new marker of pigment cell precursors during mouse embryonic development, Mech. Dev., 116, 209-212 (2002).
10. Atlas of Genetics and Cytogenetics in Oncology and Haematology.
11. A. Taïeb and M. Picardo, "Epidemiology, definition, and classification" in Vitiligo, Eds. M. Picardo and A. Taïeb, Springer-Verlag: Berlin (2010).
12. S.A. Birlea, R.A. Spritz, and D.A. Norris, "Vitiligo" in Fitzpatrick's Dermatology in General Medicine, 8th ed., Eds. L.A. Goldsmith, S.I. Katz, B.A. Gilchrest, A.S. Paller, D.J. Leffell, and K. Wolff, McGraw-Hill: New York (2012).
13. E.H. Kemp, S. Emhemad, D.J. Gawkrodger, and A.P. Weetman, "Autoimmunity in vitiligo" in Autoimmune Disorders-Pathogenetic Aspects, Ed. C.P. Mavragani, InTech Press: Rijeka, Croatia (2011).
14. J.A. Mosenson, J.M. Eby, C. Hernandez, I.C. Le Poole, A central role for inducible heat-shock protein 70 in autoimmune vitiligo, Exp. Dermatol., 22, 566-569 (2013).
15. M. Rendon, M. Berneburg, I. Arellano, and M. Picardo, Treatment of melasma, J. Am. Acad. Dermatol., 54, S272-S281 (2006).
16. G.A. Johnston, L. Sviland, and J.G.A. McLelland, Melasma of the arms associated with hormone replacement therapy, Br. J. Dermatol., 139, 932 (1998).
17. E.C. Davis and V.D. Callender, Postinflammatory hyperpigmentation: a review of the epidemiology, clinical features, and treatment options in skin of color, J. Clin. Aesthet. Dermatol., 3, 20-31 (2010).
18. A.G. Pandya and I.L. Guevara, Disorders of hyperpigmentation, Dermatol. Clin., 18, 91-98 (2000).
19.
D. Hexsel, I. Arellano I, and M. Rendon, Ethnic considerations in the treatment of Hispanic and Latin-American patients with hyperpigmentation, Br. J. Dermatol., 156S, 7-12 (2004).
20. N.L. Lacz, J. Vafaie, N.I. Kihiczak, and R.A Schwartz, Postinflammatory hyperpigmentation: a common but troubling condition, Int. J. Dermatol., 43, 362-365 (2004).
21. A.J. Stratigos and A.D. Katsambas, Optimal management of recalcitrant disorders of hyperpigmentation in dark-skinned patients, Am. J. Clin. Dermatol., 5, 161-168 (2004).
22. R.M. Halder and J.J. Nordlund, "Topical treatment of pigmentary disorders" in The Pigmentary System: Physiology and Pathophysiology, Eds. J.J. Nordlund, R.E. Boissy, V.J. Hearing, R.A. King, W.S. Oetting, and J.P. Ortonne, 2nd ed., Blackwell Publishing: Malden, MA (2006).
23. M. Yaar and B.A. Gilchrest, "Aging of the skin" in Fitzpatrick’s Dermatology in General Medicine, 6th ed., Eds. I.M. Freedberg, A.Z. Eisen, K. Wolff, K. F. Austen, L.A. Goldsmith, and S.I. Katz, McGraw-Hill: New York (2003).
24. J.J. Nordlund, R.E. Boissy, V.J. Hearing, R.A. King, W.S. Oetting, and J.-P. Ortonne, The Pigmentary System: Physiology and Pathophysiology, 2nd ed., Oxford Univ. Press: New York (2006).
25. M.B. Rozanowska, "Properties and functions of ocular melanins and melanosomes" in Melanins and Melanosomes: Biosynthesis, Biogenesis, Physiological, and Pathological Functions, Eds. J. Borovanský and P.A. Riley, Wiley-Blackwell: Weinheim (2011).
26. Z. Abdel-Malek, V.B. Swope, and J.J. Nordlund, The nature and biological effects of factors responsible for proliferation and differentiation of melanocyte, Pigment Cell Res., 3, 43-47 (1992).
27. Z.A. Abdel-Malek, V.B. Swope, J. Pallas, K. Krug, and J.J. Nordlund, Mitogenic, melanogenic, and cAMP responses of cultured neonatal human melanocytes to commonly used mitogens, J. Cell. Physiol., 150, 416-425 (1992).
28. R. Halaban, The regulation of normal melanocyte proliferation, Pigment Cell Res., 13, 4-14 (2000).
29. G. Prota, Melanins and Melanogenesis, Academic Press: New York (1992).
30. S. Ito, K. Wakamatsu, and H. Ozeki, Chemical analysis of melanins and its application to the study of the regulation of melanogenesis, Pigment Cell Res., 13, 103-109 (2000).
31. Y. Yamaguchi and V.J. Hearing, "Melanocyte distribution and function in human skin: effects of UV radiation" in From Melanocytes to Malignant Melanoma, Eds. V.J. Hearing and S.P.L. Leong, Humana Press: Totowa, NJ (2005).
32. Y. Yamaguchi, K.Takahashi, B.Z. Zmudzka, A. Kornhauser, S.A. Miller, T. Tadokoro, W. Berens, J.Z. Beer, and V.J. Hearing, Human skin responses to UV radiation: pigment in the upper epidermis protects against DNA damage in the lower epidermis and facilitates apoptosis, FASEB J., 20, 1486-1488 (2006).
33. J.L. Rees, The genetics of sun sensitivity in humans, Am. J. Hum. Gen., 75, 739-751 (2004).
34. K. Wakamatsu, R. Kavanagh, A.L. Kadekaro, S. Terzieva, R.A. Sturm, S. Leachman, Z. Abdel-Malek, and S. Ito, Diversity of pigmentation in cultured human melanocytes is due to differences in the type as well as quantity of melanin, Pigment Cell Res., 19, 154-162 (2006).
35. Y. Miyamura, S.G. Coelho, R. Wolber, S.A. Miller, K. Wakamatsu, B.Z. Zmudzka, S. Ito, C. Smuda, T. Passeron, W. Choi, J. Batzer, Y. Yamaguchi, J.Z. Beer, and V.J. Hearing, Regulation of human skin pigmentation and responses to ultraviolet radiation, Pigment Cell Res., 20, 2-13 (2007).
36. Y. Yamaguchi, M. Brenner, and V.J. Hearing, The regulation of skin pigmentation, J. Biol. Chem., 282, 27557-27561 (2007).
37. S. Ito and K. Wakamatsu, "Chemistry of melanins" in The Pigmentary System: Physiology and Pathophysiology, 2nd ed., Eds. J.J. Nordlund, R.E. Boissy, V.J. Hearing, R.A. King, W.S. Oetting, and J.-P. Ortonne, Oxford Univ. Press: New York (2006).
38. Y. Kondo and V.J. Hearing, Update on the regulation of mammalian melanocyte function and skin pigmentation, Expert Rev. Dermatol., 6, 97-108 (2011).
39. S. Ito and K. Wakamatsu, Chemistry of mixed melanogenesis—pivotal roles of dopaquinone, Photochem. Photobiol., 84, 582-592 (2008).
40. M. Hara, M. Yaar, and B.A. Gilchrest, Endothelin-1 of keratinocyte origin is a mediator of melanocyte dendricity, J. Invest. Dermatol., 105, 744-748 (1995).
41. R. Busca and R. Ballotti, Cyclic AMP: A key messenger in the regulation of skin pigmentation, Pigment Cell Res., 13, 60-69 (2000).
42. T. Tadokoro, Y. Yamaguchi, J. Batzer, S.G. Coelho, B.Z. Zmudzka, S.A. Miller, R. Wolber, J.Z. Beer, and V.J. Hearing, Mechanisms of skin tanning in different racial/ethnic groups in response to ultraviolet radiation, J. Invest. Dermatol., 124, 1326-1332 (2005).
43. T.S. Kuroda and M. Fukuda, Rab27A-binding protein Slp2-a is required for peripheral melanosome distribution and elongated cell shape in melanocytes, Nat. Cell Biol., 6, 1195-1203 (2004).
44. Y. Yamaguchi and V.J. Hearing, Physiological factors that regulate skin pigmentation, Biofactors, 35, 193-199 (2009).
45. T. Kushimoto, J.C. Valencia, G.E. Costin, K. Toyofuku, H. Watabe, K. Yasumoto, F. Rouzaud, W.D. Vieira, and V.J. Hearing, The Seiji memorial lecture: the melanosome: an ideal model to study cellular differentiation, Pigment Cell Res., 16, 237-244 (2003).
46. K. Yasumoto, H. Watabe, J.C. Valencia, T. Kushimoto, T. Kobayashi, E. Appella, and V.J. Hearing, Epitope mapping of the melanosomal matrix protein gp100 (PMEL17): rapid processing in the endoplasmic reticulum and glycosylation in the early Golgi compartment, J. Biol. Chem., 279, 28330-28338 (2004).
47. T. Hoashi, H. Watabe, J. Muller, Y. Yamaguchi, W.D. Vieira, and V.J. Hearing, Mart-1 is required for the function of melanosomal matrix protein PMEL17/GP100 and the maturation of melanosomes, J. Biol. Chem., 280, 14006-14016 (2005).
48. W. Choi, L. Kolbe, and V.J. Hearing, Characterization of the bioactive motif of neuregulin-1, a fibroblast-derived paracrine factor that regulates the constitutive color and the function of melanocytes in human skin. Pigment Cell Melanoma Res. 25, 1-5 (2012).
49. Y. Yamaguchi, S. Itami, H. Watabe, K. Yasumoto, Z.A. Abdel-Malek, T. Kubo, F. Rouzaud, A. Tanemura, K. Yoshikawa, and V.J. Hearing, Mesenchymal-epithelial interactions in the skin: Increased expression of dickkopf1 by palmoplantar fibroblasts inhibits melanocyte growth and differentiation, J. Cell Biol., 165, 275-285 (2004).
50. Y. Yamaguchi, T. Passeron, H. Watabe, K. Yasumoto, F. Rouzaud, T. Hoashi, and V.J. Hearing, The effects of dickkopf 1 on gene expression and Wnt signaling by melanocytes: mechanisms underlying its suppression of melanocyte function and proliferation, J. Invest. Dermatol., 127, 1217-1225 (2007b).
51. Y. Yamaguchi, T. Passeron, T. Hoashi, H. Watabe, F. Rouzaud, K. Yasumoto, T. Hara, C. Tohyama, I. Katayama, T. Miki, and V.J. Hearing, Dickkopf 1 (DKK1) regulates skin pigmentation and thickness by affecting Wnt/β-catenin signaling in keratinocytes, FASEB J., 22, 1009-1020 (2008).
52. G.E. Costin and V.J. Hearing, Human skin pigmentation: melanocytes modulate skin color in response to stress, FASEB J., 21, 976-994 (2007).
53. T. Tadokoro, F. Rouzaud, S. Itami, V.J. Hearing, and K. Yoshikawa, The inhibitory effect of androgen and sex-hormone-binding globulin on the intracellular cAMP level and tyrosinase activity of normal human melanocytes, Pigment Cell Res., 16, 190-197 (2003).
54. A. Slominski, D.J. Tobin, S. Shibahara, and J. Wortsman, Melanin pigmentation in mammalian skin and its hormonal regulation, Physiol Rev., 84, 1155-1228 (2004).
55. Y. Lu, W.Y. Zhu, C. Tan, G.H. Yu, and J.X. Gu, Melanocytes are potential immunocompetent cells: evidence from recognition of immunological characteristics of cultured human melanocytes, Pigment Cell Res., 15, 454-460 (2002).
56. I. Tam and K. Stępień, Melanocytes—immunocompetent pigment cells, Postep. Derm. Alergol., 4, 188-193 (2007).
57. D.J. Tobin, The cell biology of human hair follicle pigmentation, Pigment Cell Melanoma Res., 24, 75-88 (2011).
58. V.J. Hearing and R.A. King, Pigmentation and Pigmentary Disorders, Part 2, Ed. L. Norman, CRC Press: Boca Raton, FL (1993).
59. G. Müller, S. Ruppert, E. Schmid, and G. Schütz, Functional analysis of alternatively spliced tyrosinase gene transcripts, EMBO J., 7, 2723-2730 (1988).
60. J.D. Laskin and L.A. Piccinini, Tyrosinase isozyme heterogeneity in differentiating B16/C3 melanoma, J. Biol. Chem., 261, 16626-16635 (1986).
61. R.L. Jolley, L.H. Evans, N. Makino, and H.S. Mason, Oxytyrosinase, J. Biol. Chem., 249, 335-345 (1974).
62. V. Ferioli, C. Rustichelli, G. Pavesi, and G. Gamberini, New combined treatment of hypermelanosis: analytical studies on efficacy and stability improvement, Int. J. Cosmet. Sci., 23, 333-340 (2001).
63. J.J.J. Fu, G.G. Hillebrand,P. Raleigh, J. Li, M.J. Marmor, V. Bertucci, P.E. Grimes, S.H. Mandy, M.I. Perez, S.H. Weinkle, and J.R. Kaczvinsky, A randomized, controlled comparative study of the wrinkle reduction benefits of a cosmetic niacinamide/peptide/retinyl propionate product regimen vs. a prescription 0.02% tretinoin product regimen, Br. J. Dermatol., 162, 647-654 (2010).
64. D. Elstein, C. Hollak, J.M. Aerts, S. van Weely, M. Maas, T.M. Cox, R.H. Lachmann, M. Hrebicek, F.M. Platt, T.D. Butters, R.A. Dwek, and A. Zimran, Sustained therapeutic effects of oral miglustat (Zavesca, N-butyldeoxynojirimicin, OGT 918) in type I Gaucher disease, J. Interit. Metab. Dis., 27, 757-766 (2004).
65. S.M. Petrescu, A.-J. Petrescu, H.N. Titu, R.A. Dwek, and F.M. Platt, Inhibition of N-glycan processing in B16 melanoma cells results in inactivation of tyrosinase but does not prevent its transport to the melanosome, J. Biol. Chem., 272, 15796–15803 (1997).
66. G. Diwakar, V. Klump, R. Lazova, and J. Pawelek, Evidence for glycosylation as a regulator of the pigmentary system: key roles of sialyl(α2-6)gal/GalNAc-terminated glycans in melanin synthesis and transfer, Glycoconj. J., 32, 413-420 (2015).
67. K. Katayama, J. Armendariz-Borunda, R. Raghow, A.H. Kang, and J.M. Seyer, A pentapeptide from type I procollagen promotes extracellular matrix production, J. Biol. Chem., 268, 9941-9944 (1993).
68. L.R. Robinson, N.C. Fitzgerald, D.G. Doughty, N.C. Dawes, C.A. Berge, and D.L. Bissett, Topical palmitoyl pentapeptide provides improvement in photoaged human facial skin, Int. J. Cosmet. Sci., 27, 155-160 (2005).
69. M.P. Lupo and A.L. Cole, Cosmeceutical peptides, Dermatol. Ther., 20, 343-439 (2007).
70. A. Abu Ubeid, L. Zhao, Y. Wang, and B.M. Hantash, Short-sequence oligopeptides with inhibitory activity against mushroom and human tyrosinase, J. Invest. Dermatol., 129, 2242-2249 (2009).
71. C.A. Natale, E.K. Duperret, J. Zhang, R. Sadeghi, A. Dahal, K.T. O'Brien, R. Cookson, J.D. Winkler, and T.W. Ridky, Sex steroids regulate skin pigmentation through nonclassical membrane-bound receptors, Elife, 5, April 26 (2017), e15104; doi: 10.7554/eLife.15104.
72. A. Gunia-Krzyżak, J. Popiół, and H. Marona, Melanogenesis inhibityors: strategies for searching for and evaluation of active compounds, Curr. Med. Chem., 23, 1-27 (2016).
73. R.J. Zauhar, G. Moyna, L. Tian, Z. Li, and W.J. Welsh, Shape signatures: a new approach to computer aided ligand- and receptor-based drug design, J. Med. Chem., 46, 5674-5690 (2003).
74. N. Ai, W.J. Welsh, U. Santhanam, H. Hu, and J. Lyga, Novel virtual screening approach for the discovery of human tyrosinase inhibitors, PLoS ONE, 9, e112788 (2014).
75. J.W. Lyga, U. Santhanam, and W.J. Welsh, Topical lightening composition and uses thereof. U.S. Patent 8,329,149 (2012).
76. E.J. Land, C.A. Ramsden, and P.A. Riley, "Toxicological aspects of melanin and melanogenesis" in The Pigmentary System: Physiology and Pathophysiology, Eds. J.J. Nordlund, R.E. Boissy, V.J. Hearing, R.A. King, W.S. Oetting, and J.P. Ortonne, 2nd ed., Blackwell Publishing: Malden, MA (2006).
77. T.C. Lei, V.M. Virador, W.D. Vieira, and V.J. Hearing, A melanocyte-keratinocyte coculture model to assess regulators of pigmentation in vitro, Anal. Biochem., 305, 260-268 (2002).
78. R. Kumar, D. Parsad, A. Kanwar, and D. Kaul, Development of melanocyte-keratincoyte co-culture model for controls and vitiligo to assess regulators of pigmentation and melanocytes, Ind. J. Dermatol. Venereol. Leprol., 78, 599-604 (2012).
79. T.J. Yoon and V.J. Hearing, Co-culture of mouse epidermal cells for studies of pigmentation, Pigm. Cell Res., 16, 159-163 (2003).
80. C.L. Cannon, P.J. Neal, J.A. Southee, J. Kubilus, and M. Klausner, New epidermal model for dermal irritancy testing, Toxicol. In Vitro, 8, 889-891 (1994).
81. G.E. Costin and H. Raabe, Optimized in vitro pigmentation screening assay using a reconstructed three dimensional human skin model, Rom. J. Biochem., 50, 15-27 (2013).
82. G.E. Costin and H. Raabe, Technical challenges associated with dosing devices used for topical treatment of three dimensional reconstructed pigmented tissues, Rom. J. Biochem., 50, 81-91 (2013).
83. F. Sahuc, B. de Wever, and M. Rosdy, Human tanned epidermis reconstituted in vitro in chemically defined medium used for evaluation of modulation of pigmentation, Pigment Cell Res., 17, 551-603, PP37 (2004).
84. EU Regulation (EC) No 1223/2009 of the European Parliament and of the Council of 30 November 2009. Official Journal of the European Union L 342/59, 59-209 (2009).
85. T.J. Yoon, T.C. Lei, Y. Yamaguchi, J. Batzer, R. Wolber, and V.J. Hearing, Reconstituted 3-dimensional human skin of various ethnic origins as an in vitro model for studies of pigmentation, Anal. Biochem., 318, 260-269 (2003).
86. J.F. Hermanns, L. Petit, C. Piérard-Franchimont, P. Paquet, G.E. Piérard, Assessment of topical hypopigmenting agents on solar lentigines of Asian women, Dermatology, 204, 281-286 (2002).
87. M. Moncrieff, S. Cotton, E. Claridge, P. Hall, Spectrophotometric intracutaneous analysis - a new technique for imaging pigmented skin lesions, Br. J. Dermatol., 146, 448-457 (2002).
88. S. Preece, S.D. Cotton, and E. Claridge, "Imaging the pigments of skin with a technique which is invariant to changes in surface geometry and intensity of illuminating light" in Proceedings of Medical Image Understanding and Analysis, Ed. D. Barber, British Machine Vision Association: Malvern, UK, 145-148 (2003).
89. P.J. Matts, P.J. Dykes, and R. Marks, The distribution of melanin in skin determined in vivo, Br. J. Dermatol., 156, 620-628 (2007).